Cerebrolysin
Porcine brain-derived peptide mixture approved in 40+ countries for stroke, TBI, and Alzheimer's disease.
🔬 Mechanism of Action
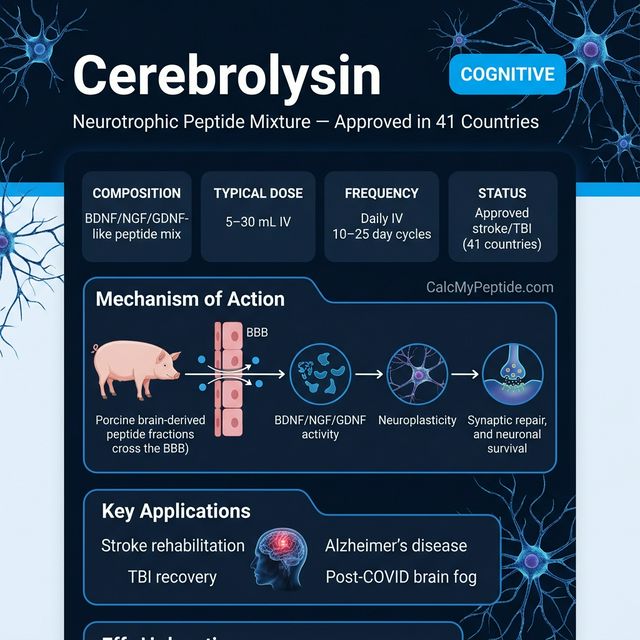
Cerebrolysin is a porcine brain-derived peptide preparation containing low-molecular-weight neuropeptides and free amino acids produced by controlled enzymatic proteolysis of lipid-free porcine brain proteins. It contains neurotrophic factors analogous to BDNF, CNTF, and GDNF.
Cerebrolysin promotes neurogenesis (new neuron formation), synaptogenesis (new synaptic connections), neuronal survival, and recovery from ischemic damage. It is approved in 40+ countries for stroke, traumatic brain injury, Alzheimer's disease, and other neurodegenerative conditions.
Source: PMID: 21281685
📜Background & History
Cerebrolysin is a neuropeptide preparation derived from purified porcine brain proteins (primarily neocortex), developed in Austria in 1949 by Ernst Ebewe. It contains a mixture of short peptides and free amino acids replicating the neurotrophic factor profile of BDNF, NGF, GDNF, IGF-1, and CNTF. Unlike recombinant growth factors that cannot cross the blood-brain barrier, Cerebrolysin's small peptide fragments do penetrate CNS tissue. It is approved in 41 countries for stroke, TBI, and dementia, with over 100 clinical trials and 40+ years of clinical use history.
🎯 Research Use Cases
- ✓Acute and chronic stroke rehabilitation (approved in 41 countries)
- ✓Traumatic brain injury (TBI) recovery
- ✓Alzheimer's disease and vascular dementia (neuroprotection)
- ✓Post-COVID cognitive impairment ("brain fog")
- ✓Performance nootropic: memory, processing speed, neuroplasticity
💉 Dosing Protocol
| Typical Dose | 5-30 mL/day (IV/IM) |
| Frequency | 1× daily for 10-20 days |
| Half-Life | ~2-4 hours (peptide mix) |
⚠️Safety & Considerations
Approved in 40+ countries. Administered IV or IM only — not subcutaneous. Generally well-tolerated. May cause headache, dizziness, or injection site reactions. Contraindicated in severe renal impairment and status epilepticus.
⚡Interactions & Contraindications
Do not mix with 5-HT containing solutions or balanced amino acid infusions. Rare: CNS stimulatory effects — reduce dose if severe sleep disruption occurs. Anti-epileptic drugs: monitor seizure threshold (both Cerebrolysin and some anti-epileptics lower threshold). Anti-MAO drugs: potential interaction with aminergic components.
🔗Synergies & Common Stacks
Cerebrolysin provides the neurotrophic factors (BDNF-like activity); Dihexa promotes the synaptogenesis HGF/MET pathway to build the synaptic connections those factors support.
Semax upregulates endogenous BDNF; Cerebrolysin delivers exogenous BDNF-like peptides. Together they comprehensively support neural repair and plasticity from multiple angles.
❓ Frequently Asked Questions
Is Cerebrolysin available in the US?▼
How is Cerebrolysin administered?▼
📖 References
- Ruther E, et al. “Cerebrolysin in vascular dementia: improvement of clinical outcome in a randomized trial.” J Neural Transm (2000). PMID: 10960558