The GLP-1 Evolution: Single → Dual → Triple Agonism
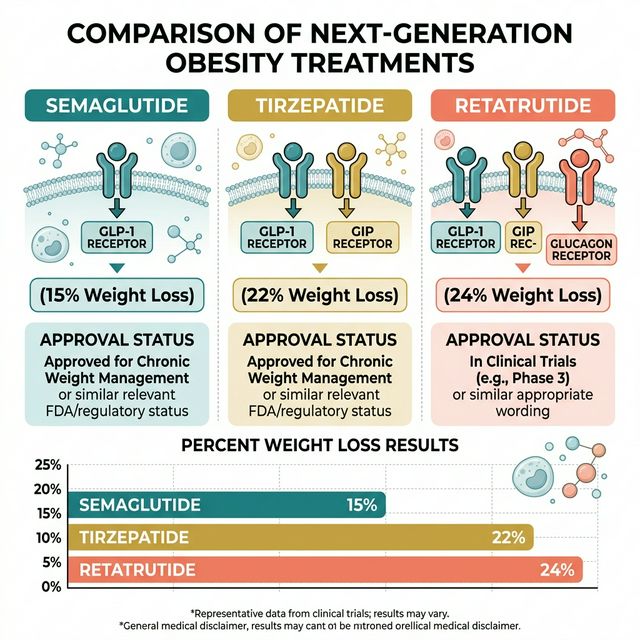
The weight loss peptide landscape in 2026 is defined by an evolution of receptor agonism. Semaglutide activates one receptor: GLP-1. Tirzepatide activates two: GLP-1 and GIP. Retatrutide activates three: GLP-1, GIP, and glucagon. Each generation produces incrementally greater weight loss — and each generation carries its own trade-offs.
This is not just a marketing story. The pharmacology is real, the clinical data is real, and the differences matter. The STEP, SURMOUNT, and TRIUMPH trial programs have collectively enrolled over 30,000 patients and produced the most robust obesity pharmacology data in medical history. Here is what the data actually says.
Semaglutide: The Single Agonist That Started Everything
Semaglutide (brand names: Ozempic for diabetes, Wegovy for weight loss) is a GLP-1 receptor agonist with 94% homology to native GLP-1. Its key modification is the C18 fatty acid chain that enables albumin binding, extending the half-life to approximately 7 days (enabling once-weekly dosing). Semaglutide activates the GLP-1 receptor on: pancreatic beta cells (enhancing insulin secretion), hypothalamic neurons (reducing appetite via POMC/CART activation), and gastric smooth muscle (delaying gastric emptying).
Clinical weight loss data: The STEP trials — the largest GLP-1 trial program — established semaglutide 2.4 mg/week as the standard of care for pharmacological obesity treatment. In STEP 1 (Wilding et al., NEJM 2021), 1,961 patients without diabetes lost an average of 14.9% of body weight at 68 weeks versus 2.4% placebo. The landmark result: ~33% of patients lost ≥20% body weight.
Dose escalation schedule (Wegovy): 0.25 mg × 4 weeks → 0.5 mg × 4 weeks → 1.0 mg × 4 weeks → 1.7 mg × 4 weeks → 2.4 mg maintenance. Slow escalation is essential to manage common GI side effects (nausea, diarrhea, vomiting) that occur in 44%, 30%, and 24% of patients respectively during escalation. Use the CalcMyPeptide GLP-1 Scheduler for your personalized week-by-week escalation plan.
Tirzepatide: The Dual Agonist That Beat Semaglutide
Tirzepatide (brand names: Mounjaro for diabetes, Zepbound for weight loss) is the first dual GIP/GLP-1 receptor agonist. It is a single peptide with engineered agonism at both receptors — not two separate peptides combined. The GIP component adds a mechanism that semaglutide lacks: GIP receptor activation on adipocytes promotes lipolysis and thermogenesis while also enhancing GLP-1's effects on insulin secretion.
Clinical weight loss data: In SURMOUNT-1 (Jastreboff et al., NEJM 2022), 2,539 patients without diabetes at the 15 mg/week maximum dose lost an average of 22.5% of body weight at 72 weeks. 36% of patients lost ≥25% body weight. In the head-to-head SURPASS-2 trial (diabetes patients), tirzepatide beat semaglutide on both A1C reduction and weight loss at all dose levels. This is the definitive head-to-head comparison.
The dose escalation for tirzepatide is: 2.5 mg × 4 weeks → 5 mg × 4 weeks → 7.5 mg × 4 weeks → 10 mg × 4 weeks → 12.5 mg × 4 weeks → 15 mg maintenance. Notably, tirzepatide appears to have slightly lower nausea rates than semaglutide at equivalent efficacy doses — possibly because the GIP component modulates GI motility differently.
Retatrutide: The Triple Agonist Setting New Records
Retatrutide (Eli Lilly, LY3437943) adds glucagon receptor agonism to the GLP-1/GIP dual mechanism. Glucagon increases energy expenditure, promotes hepatic fat oxidation, and contributes to satiety — mechanisms that are additive to GLP-1 and GIP effects. The concern with glucagon is hyperglycemia, but in retatrutide, the GLP-1 component counterbalances glucagon's glycemic effect.
Clinical weight loss data: In the Phase II trial (Jastreboff et al., NEJM 2023), 338 patients at 12 mg/week lost an average of 24.2% of body weight at 48 weeks. Extrapolated modeling suggests the plateau has not been reached — weight loss was still continuing at study end. Phase III (TRIUMPH program) is ongoing in 2026 with data expected by late 2026.
The critical caveat: retatrutide Phase II data looks extraordinary, but the sample size is small (338 patients total across all doses) compared to semaglutide's STEP program (5,000+) and tirzepatide's SURMOUNT program (5,000+). Phase III data will determine whether the results replicate at scale. Retatrutide is not yet FDA-approved; compounded versions are available through research channels.
Head-to-Head Comparison: What the Data Actually Shows
Weight loss at maximum well-studied dose: Semaglutide 2.4 mg → 14.9% (STEP 1). Tirzepatide 15 mg → 22.5% (SURMOUNT-1). Retatrutide 12 mg → 24.2% (Phase II). Direct comparison across trials carries methodological caveats (different patient populations, trial designs, durations), but the trend is clear.
GI side effects: Similar rates across all three (nausea 40-50% during escalation). Tirzepatide may have slightly lower nausea. Retatrutide Phase II nausea rates were 27% at 12 mg (lower than expected). All three see dramatic side effect reduction with slow escalation protocols.
Unique benefits by compound: Semaglutide has the strongest cardiovascular outcome data (SELECT trial: 20% reduction in MACE). Tirzepatide has the best head-to-head efficacy data versus semaglutide (SURPASS-2). Retatrutide has the highest absolute weight loss percentage but in smaller studies. All three require dose escalation — the CalcMyPeptide GLP-1 Scheduler supports all three compounds.
Which Should You Choose in 2026?
If cost and access are your primary concerns: compounded semaglutide remains the most affordable and accessible GLP-1 option. The molecule is well-understood, the dose escalation protocol is established, and compounding pharmacies offer it at approximately $100-300/month versus $1,000-1,600/month for brand Wegovy.
If maximum efficacy is your goal: tirzepatide (Mounjaro/Zepbound) has the strongest head-to-head data against semaglutide and is FDA-approved with a robust safety database. Compounded tirzepatide is also increasingly available.
If you want the cutting edge: retatrutide is producing the most impressive weight loss numbers, but it is not yet FDA-approved and the Phase III safety profile is incomplete. It is available through research peptide channels and some compounding pharmacies.
Important: All three require medical supervision, metabolic monitoring (fasting glucose, lipid panel, liver function), and a structured dose escalation protocol. These are potent metabolic drugs, not casual supplements. Use the CalcMyPeptide calculators for reconstitution math and dose scheduling, but work with a qualified healthcare provider for protocol design and monitoring.
