Oral vs Injectable Peptides: Why Bioavailability Matters
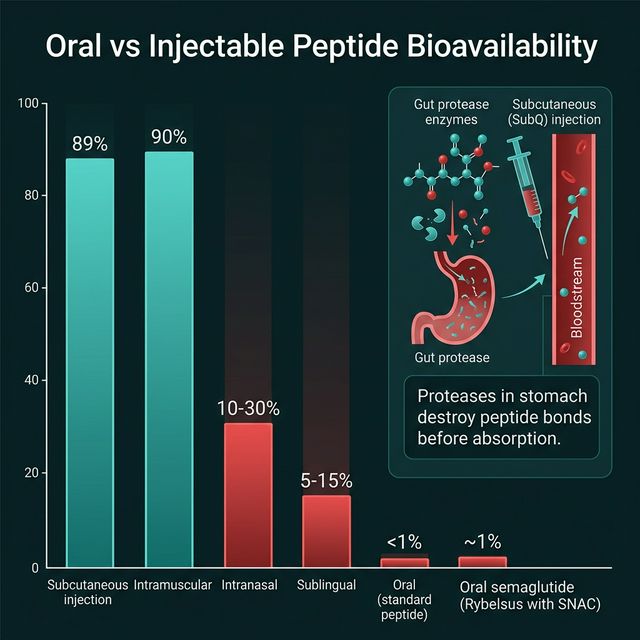
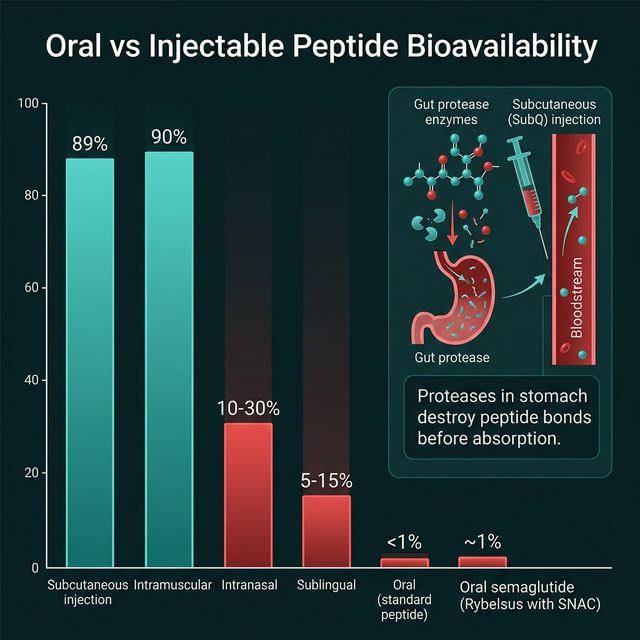
Bioavailability is the fraction of an administered dose that reaches systemic circulation in its active form. For an injectable peptide given subcutaneously, bioavailability approaches ~89-100% — nearly the full dose enters the bloodstream. For an oral peptide, bioavailability is typically 0-1% because the gastrointestinal tract destroys peptides with extreme efficiency: stomach acid hydrolyzes peptide bonds at low pH, and pancreatic proteases (trypsin, chymotrypsin, elastase) cleave virtually every exposed cleavage site in a peptide before it can be absorbed. Only specialized delivery systems, unusual molecular stability, or local (not systemic) targets allow oral peptides to be clinically meaningful (Drucker DJ, Nat Rev Drug Discov, 2020, PMID: 31434983). CalcMyPeptide provides dose calculators for both subcutaneous and oral peptide preparations.
Why Are Most Peptides Injected?
Three GI barriers destroy oral peptides before they can be absorbed:
1. Gastric acid (pH 1.5–3.5): Protonates peptide bonds, promoting hydrolysis (chemical cleavage). Most peptides begin degrading within minutes of reaching the stomach.
2. Proteolytic enzymes: The pancreas secretes trypsin, chymotrypsin, and elastase into the small intestine. These enzymes cleave peptides at specific amino acid sequences with high efficiency — designed by evolution to digest dietary protein completely into free amino acids.
3. Intestinal epithelial barrier: Even if a peptide fragment survives acids and enzymes, the tight junctions of the intestinal epithelium exclude molecules >500 Da (daltons). Most therapeutic peptides are 500-5,000 Da — too large for passive paracellular absorption.
The combined effect: oral bioavailability for most unprotected peptides is effectively 0%. Semaglutide, with the most sophisticated oral peptide delivery system (SNAC), achieves only ~1% bioavailability (Aroda VR et al., Diabetes Care, 2019, PMID: 30820234).
Oral Peptides That Work: The Special Cases
| Peptide | Oral BA | Mechanism | Clinical Use |
|---|---|---|---|
| Semaglutide (Rybelsus) | ~1% | SNAC delivery system | T2D (FDA-approved) |
| BPC-157 | ~5-10%* | Protease-resistant structure | GI-specific effect only |
| Cyclosporine | 30% | Lipophilic; resists degradation | Immunosuppression |
| Desmopressin (DDAVP) | ~0.1-1% | Intranasal used instead | Diabetes insipidus |
*BPC-157 oral bioavailability for GI-local effect only; systemic bioavailability for non-GI targets essentially 0%.
SNAC mechanism for semaglutide (Davies M et al., Br J Clin Pharmacol, 2021, PMID: 33159350): SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) creates a localized pH increase in the gastric mucosa adjacent to the tablet, inhibiting pepsin activity and promoting transcellular absorption through the gastric epithelium — bypassing the enzyme-rich small intestine entirely.
Injectable Peptide Bioavailability by Route
| Route | Bioavailability | Peak Time | Notes |
|---|---|---|---|
| Subcutaneous (SubQ) | 85-100% | 1-8 hours (peptide-dependent) | Standard for most peptides |
| Intramuscular (IM) | 85-100% | 30 min-4 hours | Faster peak; more discomfort |
| Intravenous (IV) | 100% | Immediate | Hospital setting; not for home use |
| Intranasal | 5-30% | 15-30 min | Semax, Selank; convenient for cognitive peptides |
| Sublingual | 5-30% | 15-45 min | Emerging; some peptide products available |
| Transdermal | <5% | 2-8 hours | Very limited; only possible for very small peptides |
For most research peptides, subcutaneous injection is the standard and most practical route. The absorption is consistent, slow, and sustained — producing a smooth pharmacokinetic curve that matches the biological effect duration preferred for most peptide protocols.
Bioavailability and Dose Calculation: Why Route Matters
Route of administration directly affects the dose required to achieve a given therapeutic effect. If semaglutide SubQ bioavailability is 89% and oral bioavailability is 1%, then to achieve the same systemic exposure via oral: the oral dose must be 89× higher. This is why Rybelsus 14 mg oral daily provides roughly the same effect as Ozempic 0.5-1 mg weekly SubQ.
For research peptides, dose recommendations in literature are virtually always stated assuming SubQ injection. If you are exploring any alternative route (intranasal, sublingual, oral), the bioavailability fraction must be factored into your dose. CalcMyPeptide's reconstitution calculator automatically handles SubQ concentration math — enter your vial strength, water volume, and target dose to get exact syringe units.
Does Oral BPC-157 Work for Gut Health?
BPC-157 is one of the only research peptides with documented stability against gastric proteolysis (Sikiric P et al., J Physiol Pharmacol, 2006, PMID: 17106110). It survives transit through the stomach and small intestine to accumulate in GI tissue — explaining why oral BPC-157 shows effects in GI-specific animal models (gastric ulcers, colitis, bowel anastomosis healing). This makes oral BPC-157 a plausible option specifically for gut health targets. However, oral BPC-157 does NOT achieve meaningful systemic bioavailability — its GI protective effects are local. For systemic effects (tendon healing, brain-gut axis, systemic anti-inflammation), subcutaneous or intramuscular injection is required.
What Is the Best Route for Research Peptides?
For most research peptides, SubQ injection is the optimal route: highest bioavailability, most consistent absorption, lowest pain, easiest self-administration. IM injection is appropriate when faster onset is clinically relevant (e.g., TB-500 loading phase, BPC-157 for acute injury). Intranasal delivery is specifically relevant for cognitive peptides (Semax, Selank) which have documented nasal bioavailability and CNS delivery via the olfactory route. Oral is only appropriate for GI-specific BPC-157 applications and pharmaceutical-grade semaglutide (Rybelsus). For all route choices, read our peptide injection master guide and use CalcMyPeptide for precise dose calculations.