Can BPC-157 Survive Your Stomach?
Every other peptide on this website gets destroyed by gastric acid and proteolytic enzymes within minutes. BPC-157 is the exception — and the science behind why it survives is genuinely fascinating.
BPC-157 (Body Protection Compound-157) is a 15-amino-acid peptide (GEPPPGKPADDAGLV) derived from human gastric juice. It is a fragment of a larger protein found naturally in the stomach. This origin is the key to its acid stability: BPC-157 evolved to function in the most acidic environment in the human body (gastric pH 1.5-3.5). Its amino acid sequence and tertiary structure confer resistance to pepsin cleavage and acid hydrolysis that other peptides simply do not have.
Sikiric et al. (multiple publications, University of Zagreb) demonstrated that oral BPC-157 in rats survived gastric transit, accumulated in gastrointestinal tissue, and produced measurable biological effects including mucosal healing, anti-inflammatory activity, and cytoprotection. The key finding: BPC-157 administered orally concentrated in the GI tract and produced local effects equivalent to parenteral dosing for gut-specific endpoints.
The Bioavailability Reality: Local vs Systemic
Here is where the nuance matters. "Surviving stomach acid" and "achieving systemic bioavailability" are not the same thing. BPC-157 survives the stomach — but how much enters the systemic circulation?
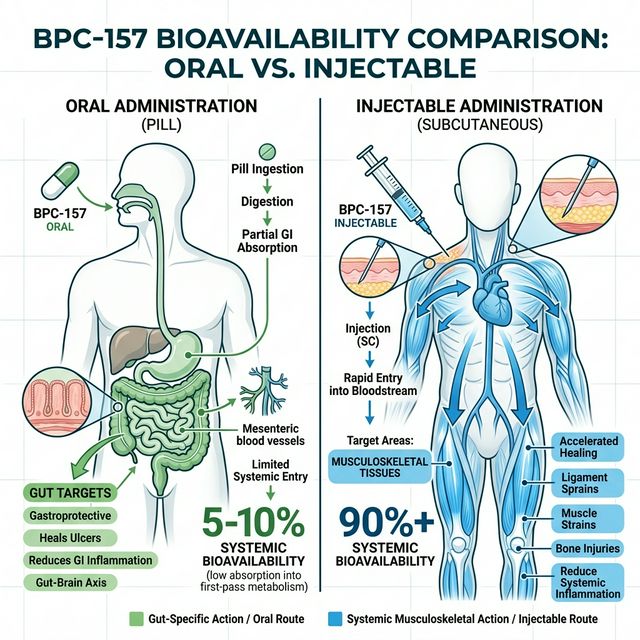
The honest answer: we do not have precise pharmacokinetic bioavailability data for oral BPC-157 in humans. The animal data suggests that oral BPC-157 produces robust local effects in the GI tract (esophageal healing, gastric ulcer protection, colitis improvement, gut motility normalization) at doses that may not achieve therapeutically relevant systemic blood levels.
For systemic targets — tendon injuries, joint pain, muscle tears, systemic inflammation — subcutaneous injection remains the established route. SubQ bioavailability for peptides is approximately 89%, meaning nearly all of the injected dose reaches systemic circulation. Oral bioavailability for BPC-157, extrapolated from animal PK studies, is estimated at 5-15% for gut tissue accumulation and <2% for systemic plasma levels. This is dramatically better than any other peptide (most are 0%), but still low for systemic therapy.
When Oral BPC-157 Makes Sense
Oral BPC-157 is pharmacologically rational for: (1) gastric ulcers and gastric mucosal healing — the peptide concentrates directly at the site of pathology; (2) esophageal damage (acid reflux, Barrett's esophagus) — same principle of local concentration; (3) inflammatory bowel conditions (ulcerative colitis, Crohn's) — BPC-157 has demonstrated mucosal healing in multiple animal models of colitis; (4) gut motility disorders — BPC-157 interacts with the enteric nervous system to normalize both hyperactive and hypoactive motility; (5) NSAID-induced GI damage — BPC-157 directly counteracts NSAID gastropathy in animal models.
In all of these cases, the target tissue is the GI tract itself. Oral delivery places the peptide directly where it needs to work. For gut-specific applications, oral BPC-157 is not a compromise — it may actually be the optimal route.
When You Need Injectable
Injectable (subcutaneous) BPC-157 is required when the target is not the GI tract. This includes: tendon and ligament injuries (rotator cuff, patellar tendon, Achilles tendon), muscle strains, joint inflammation, post-surgical wound healing, nerve injury recovery, and systemic anti-inflammatory effects. For these targets, you need BPC-157 in the bloodstream at therapeutically relevant concentrations, and oral dosing does not reliably deliver that.
Standard injectable protocol: 250-500 mcg BPC-157 subcutaneously, 1-2 times daily, injected near the injury site or into the abdominal subcutaneous tissue. For a 5 mg vial reconstituted with 2 mL bacteriostatic water: concentration = 2,500 mcg/mL. For 250 mcg: 10 units on a U-100 syringe. Duration: typically 4-8 weeks for injury recovery.
The practical recommendation: if your target is the gut, try oral BPC-157 first (typical dose: 250-500 mcg in capsule form, taken on an empty stomach). If your target is a musculoskeletal injury or systemic recovery, use subcutaneous injection. Some practitioners use both routes simultaneously — oral for GI protection (especially if using NSAIDs) and injectable for injury recovery. Use the CalcMyPeptide reconstitution calculator for injectable dosing math.