What Is Retatrutide?
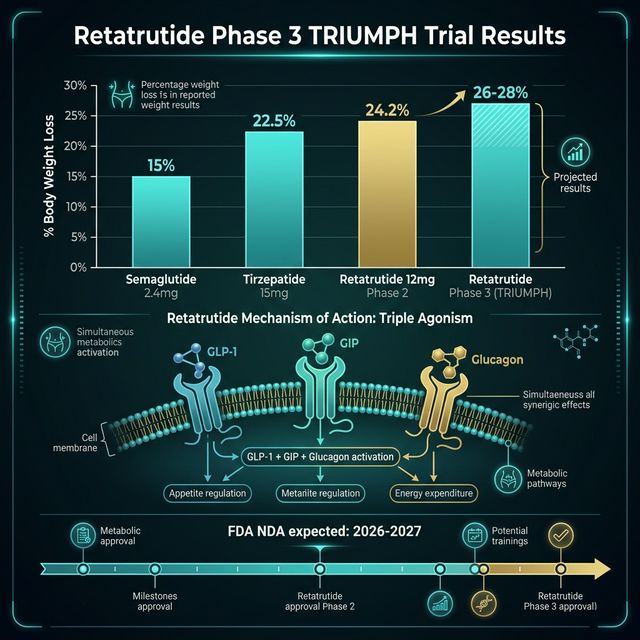
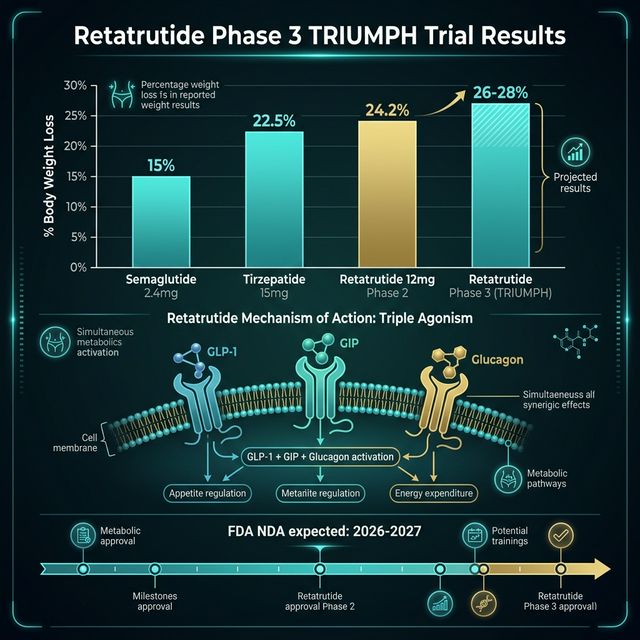
Retatrutide (LY3437943) is an investigational peptide agonist that simultaneously activates three hormone receptors: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors. This triple-agonist mechanism distinguishes it from semaglutide (GLP-1 only) and tirzepatide (GLP-1 + GIP dual agonist). Eli Lilly developed retatrutide as a next-generation obesity and metabolic disease treatment. Phase 2 data published in 2023 showed mean weight loss of up to 24.2% over 48 weeks — surpassing all previously published clinical results for any obesity drug (Jastreboff AM et al., N Engl J Med, 2023, PMID: 37351564). CalcMyPeptide provides free GLP-1 dose calculators applicable to all GLP-1 class peptides.
Retatrutide Phase 2 Results: Weight Loss Data
The retatrutide Phase 2 trial (n=338) tested doses from 1 mg to 12 mg weekly by subcutaneous injection — the same route and interval as semaglutide and tirzepatide. At 48 weeks:
| Dose | Mean Weight Loss | % of Participants with ≥15% Weight Loss |
|---|---|---|
| 4 mg/week | 17.5% | ~60% |
| 8 mg/week | 22.8% | ~75% |
| 12 mg/week | 24.2% | ~83% |
| Placebo | 2.1% | ~5% |
These figures represent the highest weight loss percentages ever reported in a pharmaceutical trial for obesity as of publication (Jastreboff AM et al., 2023). For context: semaglutide 2.4 mg/week achieved ~15% (STEP 1), tirzepatide 15 mg/week achieved ~21% (SURMOUNT-1), and retatrutide 12 mg/week achieved ~24%.
How Does the Triple-Agonist Mechanism Work?
Retatrutide activates three receptors with different metabolic roles:
1. GLP-1 receptor: Reduces appetite via hypothalamic signaling, slows gastric emptying, increases insulin secretion, decreases glucagon. Shared with semaglutide and tirzepatide.
2. GIP receptor: Enhances nutrient-stimulated insulin secretion, reduces post-meal glucagon via a mechanism additive to GLP-1. GIP receptor activation may also reduce the GI side effects seen with GLP-1 alone — potentially explaining tirzepatide's better tolerability vs semaglutide (Nogueiras R et al., Nat Rev Endocrinol, 2023).
3. Glucagon receptor: Stimulates hepatic fat oxidation (fat burning in the liver), increases basal metabolic rate, and reduces liver fat (hepatic steatosis). This is the new mechanism retatrutide adds beyond tirzepatide — glucagon activation drives additional energy expenditure independent of eating behavior.
The combination creates three synergistic pathways: eating less (GLP-1), processing food better (GIP), and burning more energy (glucagon) — simultaneously. This explains the superior weight loss seen in Phase 2.
Retatrutide vs Semaglutide vs Tirzepatide: Full Comparison
| Property | Semaglutide (Wegovy) | Tirzepatide (Zepbound) | Retatrutide |
|---|---|---|---|
| Receptors | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Best phase weight loss | ~15% (STEP 1) | ~21% (SURMOUNT-1) | ~24% (Phase 2) |
| Max weekly dose | 2.4 mg | 15 mg | 12 mg (Phase 2) |
| FDA approval | Yes (obesity) | Yes (obesity) | Not yet (Phase 3) |
| Primary differentiator | First approved weekly GLP-1 | Dual agonist; superior to sema | Triple agonist; added glucagon EE |
| GI side effects | Moderate-severe | Moderate | Similar to tirzepatide |
EE = Energy expenditure. Data as of 2026. Phase 3 trials ongoing.
Retatrutide Phase 3 Trial Status (2026 Update)
As of 2026, retatrutide is in Phase 3 trials under the TRIUMPH program. Key trials include:
• TRIUMPH-1: Retatrutide vs placebo for chronic weight management (primary endpoint: % weight loss at 72 weeks)
• TRIUMPH-2: Retatrutide in type 2 diabetes (primary endpoint: HbA1c reduction at 52 weeks)
• TRIUMPH-4: Open-label extension study evaluating 3-year safety and weight maintenance
The Phase 3 primary readout is expected in 2026-2027, with potential FDA filing in 2027. Eli Lilly has stated retatrutide is on the critical regulatory path following tirzepatide's commercial success.
Investor context: If retatrutide Phase 3 data replicate Phase 2 weight loss results, it could become the highest-efficacy weight management drug approved by FDA, potentially surpassing tirzepatide's commercial position.
What Is the FDA Approval Timeline for Retatrutide?
Based on Phase 3 enrollment timelines and typical FDA review periods: Phase 3 trial completion is projected for late 2026 to mid-2027. FDA NDA (New Drug Application) submission could occur in late 2027. FDA review (standard 12 months) would place potential approval in late 2028 or early 2029. This is an estimate — FDA timelines depend on data completeness, priority review eligibility, and any safety signals requiring additional study.
Breakthrough therapy designation: Retatrutide received FDA Breakthrough Therapy designation for obesity in 2023, which provides more intensive FDA guidance and review — potentially accelerating the timeline by several months.
For dose calculations on any GLP-1 class peptide, use the CalcMyPeptide GLP-1 scheduler — applicable to all weekly subcutaneous GLP-1 agonists.
How Does Retatrutide's Glucagon Component Cause Weight Loss?
Glucagon receptor activation has historically been considered undesirable from a metabolic standpoint — glucagon raises blood sugar by promoting hepatic glucose release. But targeted glucagon agonism in a triple combination changes this risk profile: GLP-1 and GIP activation simultaneously suppress glucagon's glycemic effects while preserving its fat-burning (lipolytic) and energy expenditure-enhancing properties.
The net result: liver fat oxidation increases (non-alcoholic fatty liver disease improvement), basal metabolic rate increases, and adipose tissue lipolysis is elevated — all without the hyperglycemic risk because GLP-1 is providing insulin stimulation and glucagon suppression in parallel. This metabolic flexibility — burning fat at rest — is the unique advantage the glucagon receptor adds to the triple combination.
Is Retatrutide Available as a Compounded Peptide?
No. As of 2026, retatrutide is not commercially available in any form — branded or compounded. It remains an investigational drug in Phase 3 trials. No licensed compounding pharmacy can legally produce a drug that has no FDA-approved or commercially available reference product. There is no shortage exemption applicable to an unapproved investigational drug. Any product claiming to be retatrutide available for purchase outside of clinical trials should be treated with extreme skepticism — it cannot be verified as authentic and carries significant safety and legal risks. Use the CalcMyPeptide GLP-1 scheduler for dose management of approved and compounded GLP-1 therapeutics while awaiting retatrutide's regulatory pathway.