PT-141
MC4R agonist FDA-approved (Vyleesi) for hypoactive sexual desire disorder in premenopausal women.
🔬 Mechanism of Action
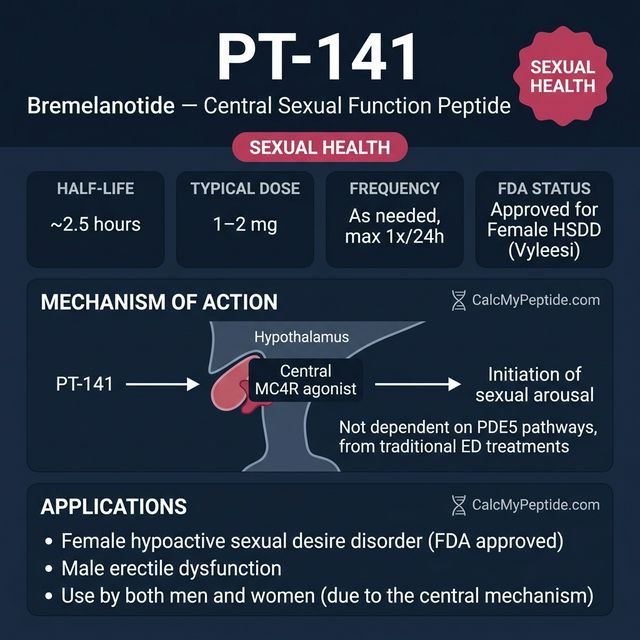
PT-141 (Bremelanotide) is a synthetic melanocortin receptor agonist that acts on MC4R (melanocortin-4 receptors) in the central nervous system to modulate sexual arousal. Unlike PDE5 inhibitors (Viagra, Cialis) that work peripherally on blood flow, PT-141 works centrally in the brain to enhance desire and arousal.
FDA-approved as Vyleesi for hypoactive sexual desire disorder (HSDD) in premenopausal women, PT-141 is also used off-label for male sexual dysfunction. It was originally derived from Melanotan II but engineered to remove the tanning effects while preserving the sexual function pathway.
Source: FDA Label (Vyleesi)
📜Background & History
PT-141 (Bremelanotide) is a cyclic heptapeptide derived from alpha-MSH (alpha-melanocyte-stimulating hormone) developed by Palatin Technologies. Unlike sildenafil and other PDE5 inhibitors that act peripherally, PT-141 acts centrally in the hypothalamus via MC4R (melanocortin-4 receptor) to initiate sexual arousal — making it effective for both men and women. FDA approved in 2019 as Vyleesi for hypoactive sexual desire disorder (HSDD) in premenopausal women — the first centrally acting sexual dysfunction treatment approved for women.
🎯 Research Use Cases
- ✓Female hypoactive sexual desire disorder (HSDD) — FDA approved (Vyleesi)
- ✓Male erectile dysfunction unresponsive to PDE5 inhibitors
- ✓Situational or psychogenic sexual dysfunction
- ✓Research: central arousal pathway modulation
💉 Dosing Protocol
| Typical Dose | 500-2000 mcg |
| Frequency | As needed (max 1×/24h, max 8×/month) |
| Half-Life | ~2.7 hours |
| Common Vial Sizes | 10 mg |
🧪 Reconstitution Example
⚠️Safety & Considerations
FDA-approved as Vyleesi. May cause nausea (most common side effect), flushing, and headache. Use maximum 1 dose per 24 hours, maximum 8 doses per month. May cause transient blood pressure elevation. Not recommended for uncontrolled hypertension.
⚡Interactions & Contraindications
Common side effect: transient nausea and flushing (MC1R-mediated). Causes transient blood pressure increase — contraindicated with cardiovascular disease, high blood pressure, or concurrent use of antihypertensives. Do not combine with PDE5 inhibitors (sildenafil, tadalafil) without medical clearance — additive blood pressure reduction risk. Maximum dosing frequency: once per 24 hours.
🔗Synergies & Common Stacks
Kisspeptin modulates GnRH/LH/FSH upstream of testosterone; PT-141 acts on downstream arousal circuitry. Together they address both hormonal and neurological dimensions of sexual function.
Oxytocin enhances emotional bonding and arousal via different receptors; PT-141 initiates desire centrally. May synergize for comprehensive sexual wellness protocols.
❓ Frequently Asked Questions
How quickly does PT-141 work?▼
What is the PT-141 dose?▼
Can men use PT-141?▼
📖 References
- Kingsberg SA, et al. “Bremelanotide for treatment of hypoactive sexual desire disorder (RECONNECT).” Obstet Gynecol (2019). PMID: 31135742
- Rosen RC, et al. “Melanocortin receptor agonists for the treatment of sexual dysfunction.” J Sex Med (2004). PMID: 15497047