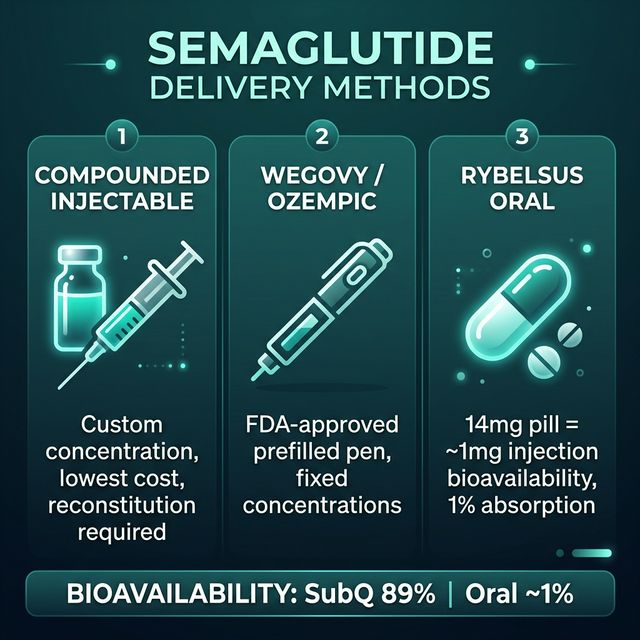
What Is the Difference Between Compounded Semaglutide and Wegovy?
Compounded semaglutide and Wegovy (branded semaglutide for weight management) contain the same active molecule — semaglutide, a GLP-1 receptor agonist. The difference is in manufacturing, form factor, regulatory status, and cost. Wegovy is produced by Novo Nordisk in its licensed pharmaceutical facilities, supplied as a pre-filled auto-injector pen in fixed doses (0.25, 0.5, 1.0, 1.7, and 2.4 mg), and carries full FDA approval for chronic weight management (Wilding JPH et al., N Engl J Med, 2021, PMID: 33567185). Compounded semaglutide is prepared by licensed 503A compounding pharmacies with a physician prescription, at custom concentrations determined by the prescriber. CalcMyPeptide's GLP-1 scheduler and reconstitution calculator work for compounded preparations of any concentration.
The pharmacological effect is identical at the same dose — both activate GLP-1 receptors in the pancreas, gut, and brain to reduce appetite and slow gastric emptying. The practical differences: Wegovy requires no reconstitution; compounded requires reconstitution math (use our calculator). Wegovy comes in a device; compounded comes in a vial. Wegovy is FDA-approved; compounded is pharmacy-prepared and carries the associated regulatory uncertainty.
How Does Oral Semaglutide (Rybelsus) Compare?
Rybelsus (oral semaglutide 7 mg or 14 mg tablet) is FDA-approved for Type 2 diabetes management and uses the same semaglutide molecule — but requires a novel oral delivery system called SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) to achieve any meaningful oral bioavailability. Without SNAC, semaglutide would be completely destroyed by GI proteases before absorption. With SNAC, it achieves approximately 1% bioavailability (Davies M et al., Br J Clin Pharmacol, 2021, PMID: 33159350).
The 1% oral bioavailability means a 14 mg oral tablet delivers the pharmacological equivalent of roughly 0.14 mg subcutaneous semaglutide — which is why the oral dose is so much higher than the injectable dose. For weight management (Wegovy-level dosing of 2.4 mg), oral delivery is not yet approved or practical — there is no oral equivalent for the higher injectable doses.
Practical guide for CalcMyPeptide users: Rybelsus requires no reconstitution. For dose math, note that 14 mg oral ≈ 1 mg SubQ in pharmacodynamic effect. Use our GLP-1 scheduler and set dose to the SubQ-equivalent if planning a switch.
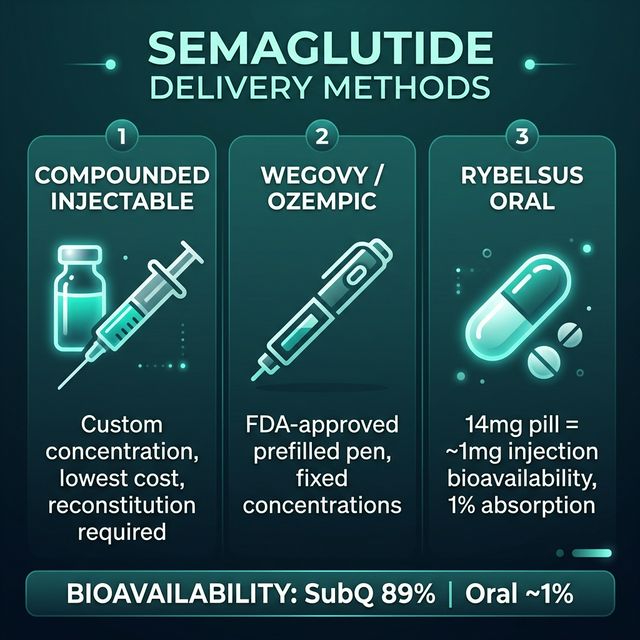
Bioavailability Comparison: SubQ vs Oral Semaglutide
| Delivery Form | Bioavailability | Dose Range | Notes |
|---|---|---|---|
| SubQ injection (Wegovy/compounded) | ~89% | 0.25–2.4 mg/week | Gold standard; standard FA (fatty acid) conjugation enables weekly dosing |
| Oral tablet (Rybelsus) | ~1% | 3, 7, 14 mg/day | Requires SNAC; take 30 min before food/water |
| Compounded injectable | ~89% | Custom | Same bioavailability as branded pen |
The subcutaneous bioavailability of 89% is documented in the Ozempic prescribing information (Kapitza C et al., J Clin Pharmacol, 2017, PMID: 28394433). This high bioavailability, combined with the fatty acid side chain that enables albumin binding, produces the 7-day half-life that makes weekly dosing possible.
Cost Comparison: Compounded vs Wegovy vs Rybelsus
| Product | List Price (Monthly) | Insurance Coverage | Notes |
|---|---|---|---|
| Wegovy 2.4 mg/week | ~$1,349 | Often covered for obesity | Manufacturer savings card available |
| Ozempic (off-label for weight) | ~$935 | Often covered for T2D only | Ozempic is T2D indicated, not weight management |
| Rybelsus 14 mg/day | ~$935 | Often covered for T2D | Not approved for weight management |
| Compounded semaglutide | $200–$400 | Usually not covered | Most patients pay out-of-pocket |
The significant cost difference explains the rapid growth of compounded semaglutide prescriptions during the Wegovy shortage period (2022–2024). Even with the shortage resolved, many patients continue using compounded formulations for cost reasons.
How to Calculate Doses for Compounded Semaglutide
Compounded semaglutide requires manual dose math because concentration varies by prescription. The formula: Units to draw = Desired dose (mg) ÷ Concentration (mg/mL) × 100.
Worked example: 5 mg semaglutide vial + 2.5 mL BAC water = concentration of 2 mg/mL. For a 0.25 mg starting dose: 0.25 ÷ 2 × 100 = 12.5 units. For a 0.5 mg dose: 0.5 ÷ 2 × 100 = 25 units. Use the CalcMyPeptide GLP-1 dose scheduler to generate your full escalation schedule with unit calculations for any vial concentration.
Or use our free Reconstitution Calculator — enter peptide = semaglutide, vial strength = 5 mg, water = 2.5 mL, and your target dose — to instantly get the exact units to draw.
Is Compounded Semaglutide as Effective as Wegovy?
Pharmacologically, compounded semaglutide is the same molecule as Wegovy semaglutide. Clinical evidence shows semaglutide 2.4 mg/week produces ~15% mean weight loss (STEP 1, Wilding JPH et al., 2021, n=1,961). Compounded semaglutide at the same dose and the same route should produce the same clinical effect — the active molecule is identical. The key variables are dose adherence, concentration accuracy of the compounded product, and purity/correct product from the compounding pharmacy.
Verification: Always request a Certificate of Analysis (CoA) with HPLC purity ≥98% and mass spectrometry molecular weight confirmation from your compounding pharmacy. Both should appear on a lot-specific document, not a generic test report.
Why Is Oral Semaglutide Dosed So Much Higher Than Injectable?
Oral bioavailability: 1% (Rybelsus). SubQ bioavailability: 89% (branded and compounded injectable). The ratio is approximately 89:1. So a 14 mg oral dose delivers the equivalent systemic exposure of approximately 0.14 mg subcutaneous — less than the lowest injectable starting dose. To deliver injectable-equivalent exposure orally, doses would need to be 89× higher than SubQ doses. For the 2.4 mg/week Wegovy dose, the oral equivalent would be approximately 213 mg — well beyond practical tablet formulation. This is why oral semaglutide is approved only for Type 2 diabetes (lower target effect) and not for the higher-dose chronic weight management indication.
What Is the FDA Status of Compounded Semaglutide in 2026?
The FDA declared the semaglutide shortage resolved in 2025, removing the shortage exemption that had allowed widespread compounding. FDA sent warning letters to compounding facilities in 2024–2025 and issued guidance restricting semaglutide compounding. As of mid-2026, 503A compounding of semaglutide is substantially restricted — pharmacies can only compound for patients with a demonstrated clinical need that cannot be met by the commercially available product (e.g., specific allergy to excipients in the branded pen). The broadly accessible compounded market has significantly contracted. For the most current status, consult an attorney or regulatory consultant specializing in compounding pharmacy. Use the CalcMyPeptide GLP-1 scheduler to optimize dosing for whichever form you are using.