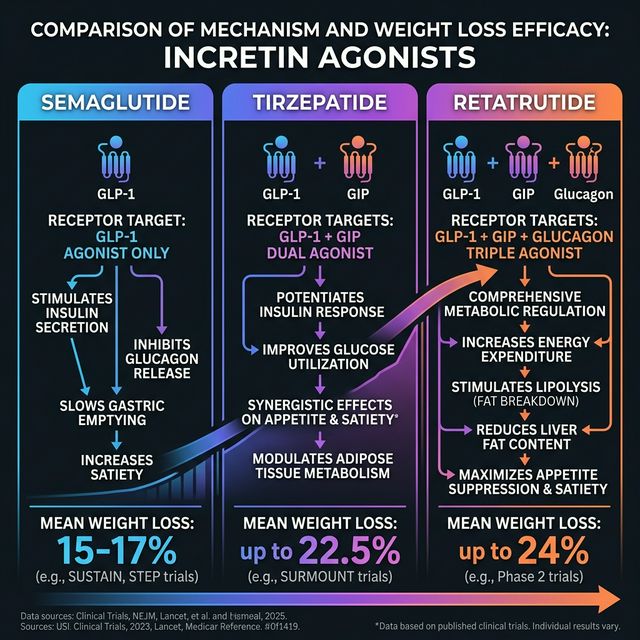
What Is Retatrutide?
Retatrutide (LY3437943) is a triple-acting agonist peptide developed by Eli Lilly that activates three receptors: GIP, GLP-1, and glucagon. It represents the next evolution beyond single-agonist (semaglutide) and dual-agonist (tirzepatide) GLP-1 therapies. The addition of glucagon receptor activation increases energy expenditure and fat oxidation beyond what GLP-1 alone achieves.
Triple Agonist Mechanism
• GLP-1 receptor: Reduces appetite, slows gastric emptying, enhances insulin secretion
• GIP receptor: Improves insulin sensitivity, promotes fat mobilization from adipose tissue
• Glucagon receptor: Increases hepatic glucose output (counterbalanced by GLP-1/GIP), increases energy expenditure, and promotes fat oxidation in the liver
The glucagon component is the key differentiator. While it might seem counterintuitive (glucagon raises blood sugar), the combined GLP-1 + GIP activity offsets this while the metabolic benefits of glucagon (increased energy expenditure) are retained.
Half-Life and Pharmacokinetics
Retatrutide has an estimated half-life of approximately 6 days (144 hours), supporting once-weekly subcutaneous dosing. Steady state is reached after approximately 4-5 weeks of consistent weekly dosing.
Phase II Dose Escalation
Based on the Phase II trial (NCT04881706), the escalation schedule for the 12 mg maintenance arm:
• Weeks 1-4: 2 mg/week
• Weeks 5-8: 4 mg/week
• Weeks 9-12: 8 mg/week
• Week 13+: 12 mg/week (maintenance)
Slower escalation is recommended for those experiencing GI side effects.
Phase II Clinical Results
In the 48-week Phase II trial (n=338), participants on the 12 mg arm achieved a mean weight loss of 24.2% from baseline, the highest weight reduction observed for any anti-obesity medication in a controlled trial at the time of publication.
HbA1c reductions of 2.02% were observed in participants with type 2 diabetes.
Source: Jastreboff AM, et al. N Engl J Med. 2023;389:514-526. DOI: 10.1056/NEJMoa2301972
Reconstitution for Compounded Retatrutide
For a compounded 10 mg vial reconstituted with 2 mL BAC water:
• Concentration = 5 mg/mL
• Using a 100-unit syringe: each unit = 0.05 mg
• For a 2 mg dose: draw 40 units
• For a 4 mg dose: draw 80 units
Use our GLP-1 Dose Scheduler to generate your complete week-by-week retatrutide schedule with syringe unit calculations.
Regulatory Status
As of 2026, retatrutide is in Phase III clinical trials. It is not yet FDA-approved. Phase III studies (TRIUMPH program) are expected to report data in late 2025-2026. Compounded versions are available through licensed pharmacies.