Why Mitochondria Are the Longevity Bottleneck
Every cell in your body (except red blood cells) contains mitochondria — organelles that produce ATP, the universal energy currency. A typical human cell contains 1,000-2,000 mitochondria. A cardiomyocyte (heart muscle cell) contains 5,000-7,000. Your body produces approximately 40 kg of ATP per day — roughly your own body weight — recycling each ATP molecule 500-700 times.
Mitochondrial dysfunction is now considered a primary driver of aging, not merely a consequence. As we age, mitochondria accumulate oxidative damage to their own DNA (mtDNA), experience declining respiratory chain efficiency, and produce increasing levels of reactive oxygen species (ROS) in a vicious cycle: damaged mitochondria produce more ROS, which causes more damage, which produces more ROS. This "mitochondrial free radical theory of aging" has been refined by modern research but remains fundamentally supported.
The clinical consequences of mitochondrial decline: reduced cellular energy (fatigue, cognitive decline, exercise intolerance), impaired cellular repair mechanisms, increased inflammation (damaged mitochondria release pro-inflammatory DAMPs), and accelerated tissue degeneration. Heart failure, neurodegenerative disease, sarcopenia, and metabolic dysfunction all have mitochondrial components.
SS-31 (Elamipretide): Direct Mitochondrial Membrane Targeting
SS-31 (D-Arg-Dmt-Lys-Phe-NH₂), also known as elamipretide or Bendavia, is a synthetic tetrapeptide that does something remarkable: it crosses the plasma membrane, penetrates into mitochondria, and concentrates on the inner mitochondrial membrane (IMM) at exactly the location where oxidative phosphorylation occurs.
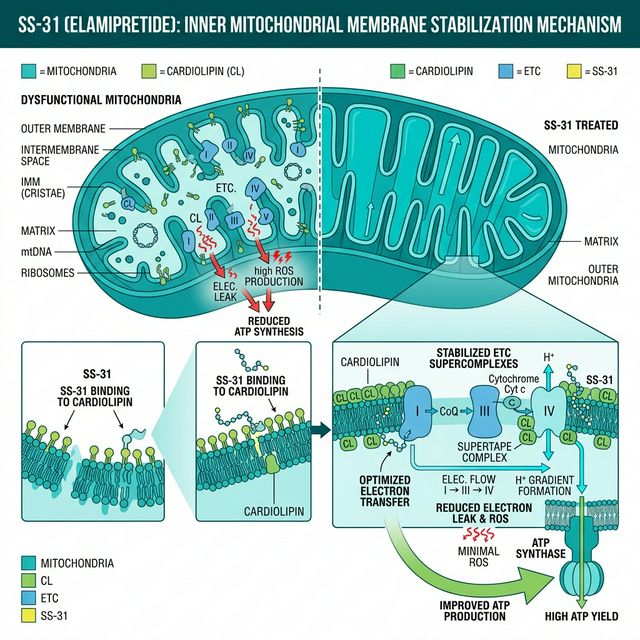
The mechanism is specific and well-characterized. SS-31 binds to cardiolipin, a phospholipid found almost exclusively on the inner mitochondrial membrane. Cardiolipin is essential for the structural organization of the electron transport chain (ETC) complexes — it acts as a molecular scaffold holding Complexes I, III, IV, and ATP synthase in their correct positions. When cardiolipin is damaged by oxidation (which accumulates with age), the ETC complexes disassemble, electron transfer becomes inefficient, and more electrons leak to form superoxide radicals.
By binding to cardiolipin, SS-31 stabilizes the ETC supercomplex organization, restores efficient electron transfer, reduces electron leak and ROS production, and improves ATP synthesis efficiency. The result: more energy production, less oxidative damage — simultaneously addressing both the cause and consequence of mitochondrial aging.
Clinical Evidence: From Heart Failure to Aging
SS-31's clinical development has focused on conditions with clear mitochondrial dysfunction: (1) Heart failure — the PROGRESS-HF and EMBRACE-HF Phase II trials evaluated elamipretide in heart failure with reduced ejection fraction (HFrEF). Results showed improvements in left ventricular end-systolic volume (LVESV) — a measure of cardiac remodeling — suggesting that mitochondrial restoration can improve heart function even in established disease.
(2) Barth syndrome — a rare genetic disease caused by mutations in tafazzin, the enzyme that remodels cardiolipin. Patients with Barth syndrome have abnormal cardiolipin, leading to severe mitochondrial dysfunction, cardiomyopathy, and skeletal myopathy. SS-31 received FDA breakthrough therapy designation for Barth syndrome, and the TAZPOWER study showed improvements in 6-minute walk distance and cardiac stroke volume.
(3) Age-related mitochondrial decline — preclinical studies in aged mice showed that SS-31 treatment restored skeletal muscle mitochondrial function, improved exercise tolerance, reduced oxidative stress markers, and rejuvenated cellular energetics to levels approaching young animals. The translational implication for human aging is significant: if mitochondrial decline is a primary driver of aging, a compound that reverses it has therapeutic potential across multiple age-related conditions.
Current Access, Dosing, and Practical Considerations
As of 2026, elamipretide has not received full FDA approval for any indication (regulatory processes are ongoing for Barth syndrome and primary mitochondrial myopathy). It is available through some compounding pharmacies (when sourced under appropriate regulatory frameworks) and through research peptide vendors.
Dosing in clinical trials: subcutaneous injection of 40 mg/day (the dose used in heart failure and Barth syndrome trials). In the optimization community, doses of 5-20 mg/day are commonly reported, though evidence for optimal dosing outside clinical trial settings is limited. Administration is subcutaneous injection, once daily.
What makes SS-31 unique among longevity interventions: it does not simply supplement a declining nutrient (like NAD+ precursors) or inhibit a damaging enzyme (like rapamycin). It physically repairs the structural foundation of energy production — the cardiolipin scaffold on the inner mitochondrial membrane. This is architectural repair, not chemical supplementation. And because mitochondrial dysfunction underlies so many age-related pathologies, a mitochondrial-targeted compound has theoretical benefits that extend across organ systems.