Gastric Survival: The Exception to the Rule
The foundational rule of clinical peptide therapy is that oral administration is ineffective because stomach acid and digestive enzymes break down amino acid chains before they can reach the bloodstream. BPC-157 is the rare exception.
BPC-157 (Body Protection Compound-157) was originally discovered in human gastric juice. It evolved specifically to operate within the extreme acidity of the stomach (pH 1.5–3.5) to protect the mucosal lining and heal ulcers. Because of this unique gastric origin, the peptide is highly stable in acidic environments and resists immediate enzymatic degradation.
The Bioavailability Dilemma
While oral BPC-157 survives the stomach, its systemic bioavailability—how much actually enters the bloodstream to treat peripheral tissues—remains a subject of intense clinical debate.
Animal models demonstrate that oral BPC-157 concentrates heavily in the gastrointestinal tract. However, its molecular weight and polarity make passive diffusion across the intestinal wall into systemic circulation highly inefficient. Estimates suggest that oral systemic bioavailability may be as low as 1% to 5%.
Conversely, subcutaneous injection bypasses the GI tract entirely, delivering nearly 100% of the peptide into the systemic circulation where it can be rapidly transported to injured connective tissues.
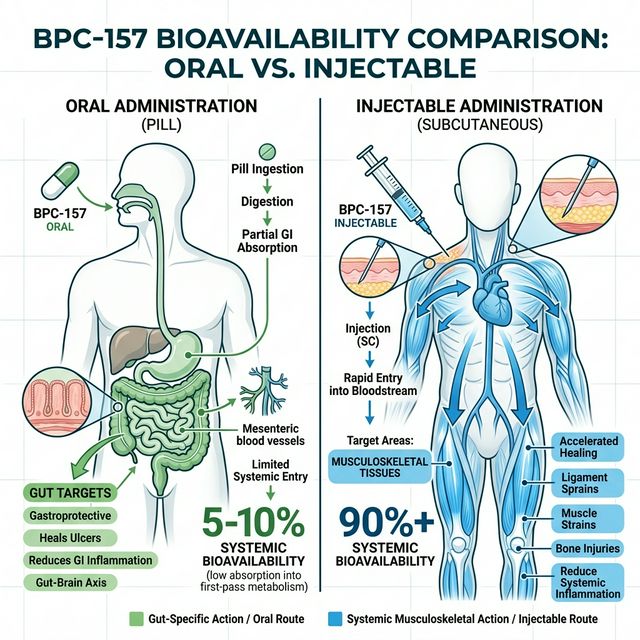
Target-Specific Administration Routes
When to use Oral BPC-157: Oral administration is indicated exclusively for gastrointestinal pathologies. If the goal is healing gastric ulcers, managing leaky gut syndrome (intestinal permeability), mitigating NSAID-induced gut damage, or addressing inflammatory bowel conditions like Crohn’s or Ulcerative Colitis, oral dosing places the peptide precisely where it is needed. Typical dosing is 250-500 mcg daily in capsule form.
When to use Injectable BPC-157: For everything else, subcutaneous injection is mandatory. If the goal is repairing a torn rotator cuff, accelerating post-surgical recovery, healing patellar tendinopathy, mitigating systemic inflammation, or neuroprotection, the tissue demands high systemic concentrations that oral delivery cannot provide. Standard protocol is 250-500 mcg subcutaneously, injected close to the site of injury if possible, 1-2 times daily.
Clinical Strategy
Do not substitute oral BPC-157 for musculoskeletal injuries simply to avoid using a needle; doing so wastes time and capital for a sub-therapeutic systemic response. Ensure the delivery route matches the tissue target. In severe, complex cases involving both systemic inflammation and severe gut permeability, advanced protocols may deploy both routes concurrently.