The 2026 Regulatory Landscape: Understanding the DQSA
The regulatory framework governing peptide therapeutics in the United States is dictated by the Drug Quality and Security Act (DQSA) of 2013, specifically Section 503A and 503B. The FDA does not "ban" or "approve" peptides arbitrarily; rather, it categorizes bulk drug substances to determine if they can be legally compounded by licensed pharmacies.
To compound a medication legally, the active pharmaceutical ingredient (API) must either be a component of an FDA-approved drug, possess a USP/NF monograph, or explicitly appear on the FDA Section 503A Bulks List.
The 503A vs 503B Divide: What Patients Must Know
The FDA strictly divides the compounding industry into two discrete legal entities:
• 503A Compounding Pharmacies: Traditional state-licensed pharmacies. They are legally restricted to compounding small, patient-specific batches based entirely on an individualized prescription from a licensed physician. They do not adhere to federal cGMP standards but must follow USP <797> guidelines for sterility.
• 503B Outsourcing Facilities: Federal FDA-registered facilities. They are permitted to manufacture large, non-patient-specific batches for "office use" by clinics and hospitals. Critically, 503B facilities must adhere to strict pharmaceutical-grade current Good Manufacturing Practice (cGMP) regulations.
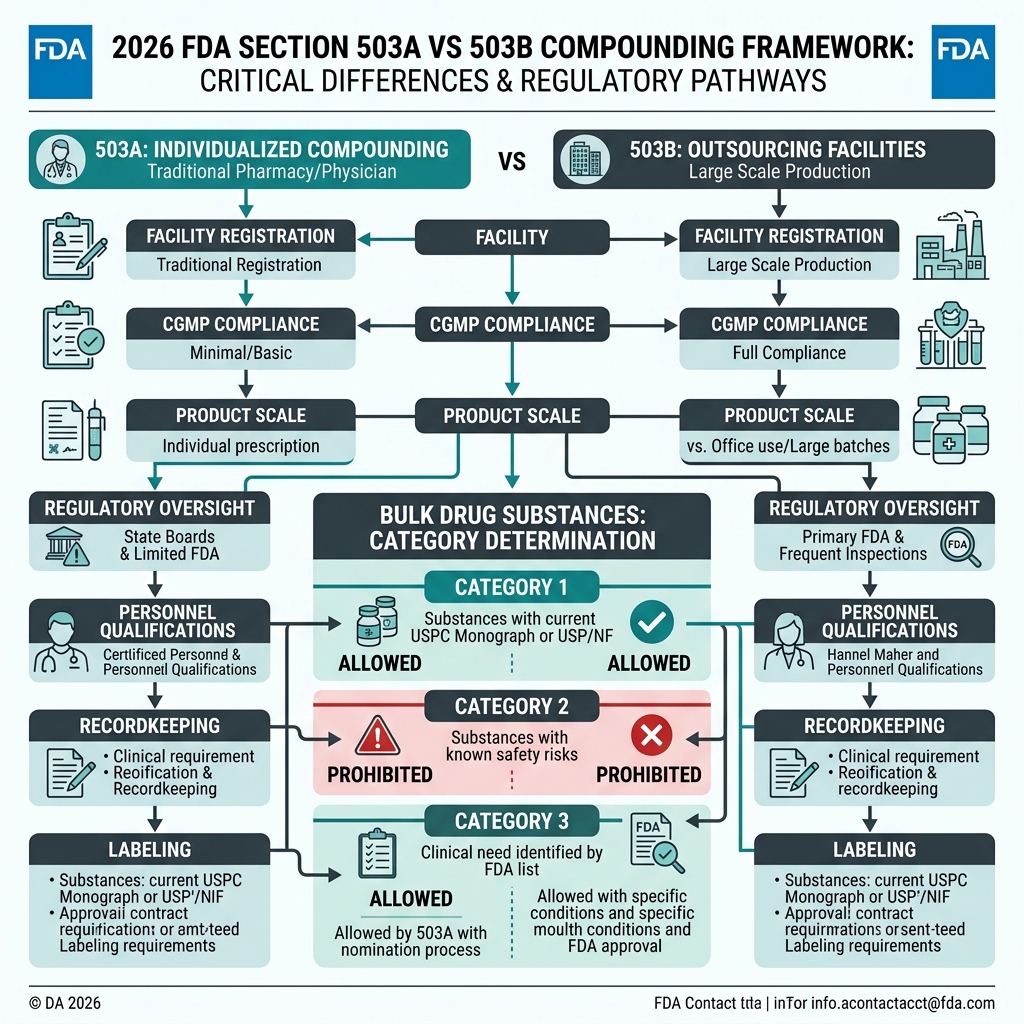
FDA Category Classifications (As of 2026)
For substances without a USP monograph or FDA approval, the FDA evaluates clinical evidence to assign them to one of three categories for compounding:
• Category 1 (Safe & Effective): The substance has sufficient clinical evidence to be safely compounded. Very few research peptides achieve this status.
• Category 2 (Under Clinical Review): The substance is actively under review by the Pharmacy Compounding Advisory Committee (PCAC). Crucially, Category 2 substances CAN STILL BE LEGALLY COMPOUNDED by 503A pharmacies until a final ruling is issued. This is the current legal harbor for leading research peptides like BPC-157, TB-500, Ipamorelin, and CJC-1295.
• Category 3 (Prohibited): The substance has been reviewed and explicitly rejected due to significant safety concerns or lack of clinical efficacy data. Pharmacies are strictly prohibited from compounding Category 3 substances.
The 2024-2026 Incretin Shortage Exemption (GLP-1s)
The most massive regulatory shift in modern peptide history involved the GLP-1 class (Semaglutide and Tirzepatide). By law, compounding pharmacies cannot create "essentially a copy" of a commercially available, FDA-approved drug (like Wegovy or Zepbound).
However, due to catastrophic supply chain failures by Novo Nordisk and Eli Lilly, the FDA added these medications to the official Drug Shortage List. This triggered an automatic statutory exemption, temporarily legalizing the compounding of generic Semaglutide and Tirzepatide to meet critical patient demand.
As of 2026, as branded supply chains have stabilized, the FDA has aggressively rolled back these shortage exemptions, issuing cease-and-desist warnings to compounders and significantly constricting the legal availability of compounded GLP-1s.
The WADA Intersection: BPC-157 and TB-500
It is vital to separate FDA compounding legality from athletic doping regulations. While BPC-157 and TB-500 currently remain in FDA Category 2 (and are thus legally accessible via 503A physician prescription), they are strictly prohibited by the World Anti-Doping Agency (WADA).
Both BPC-157 and Thymosin Beta-4 fragments are classified under WADA Section S2 (Peptide Hormones, Growth Factors, Related Substances, and Mimetics) due to their profound systemic regeneration capabilities, rendering them illegal for use by any tested competitive athlete.