⚕️ Medical Disclaimer
⚕️ Medical Disclaimer: This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any peptide.
The Analytical Necessity of Peptide Purity
In the realm of peptide therapeutics, purity is not merely a quality control metric — it is the fundamental determinant of safety and clinical efficacy. A peptide marketed as "98% pure" inherently contains 2% impurities. These impurities are not benign inert fillers; they are often truncated amino acid sequences (deletion sequences where one or more amino acids are missing from the chain), oxidized methionine or tryptophan residues, diketopiperazine cyclization products, deamidation artifacts, or synthetic byproducts such as trifluoroacetic acid (TFA) salts and residual organic solvents (DMF, NMP, DCM).
At therapeutic dosages — particularly with daily administration over multi-week protocols — the cumulative biological impact of these impurities becomes clinically significant. Truncated sequences may act as partial agonists or outright antagonists at the target receptor, potentially blocking the therapeutic effect of the intended peptide. Oxidized variants can trigger immunogenic responses, producing injection site granulomas, systemic allergic reactions, or paradoxical immune activation. TFA counter-ions, while generally considered safe at trace levels, contribute to metabolic acidosis at scale. Therefore, the ability to interpret analytical testing data — specifically High-Performance Liquid Chromatography (HPLC), Mass Spectrometry (MS), and bacterial endotoxin assays — is an absolute requirement for any informed researcher or clinician.
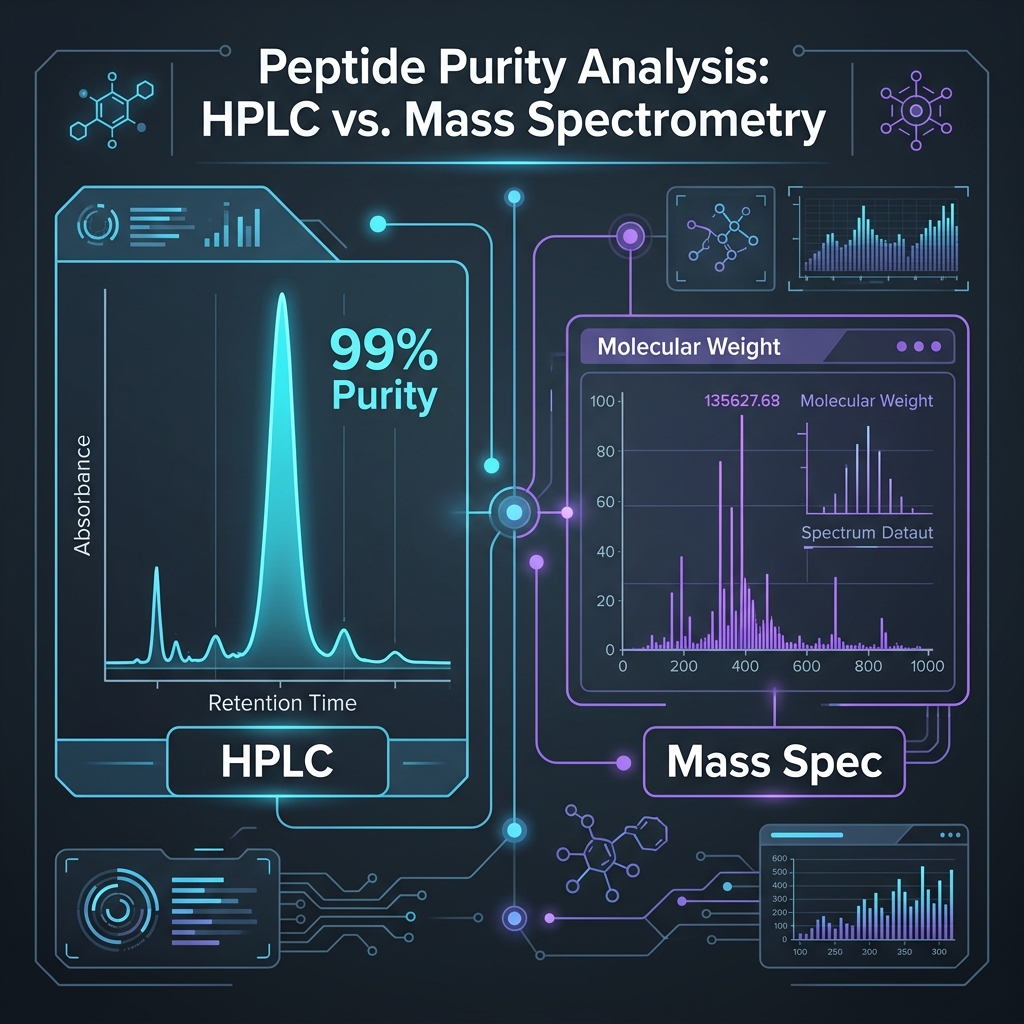
High-Performance Liquid Chromatography (HPLC): Defining Purity
HPLC is the gold standard methodology for quantifying peptide purity. The technique separates a synthesized sample into its constituent molecular components based on their differential interaction with a stationary phase — typically a C18 reversed-phase silica column — under controlled gradient elution conditions using acetonitrile/water mobile phases with 0.1% TFA modifier.
INTERPRETING THE CHROMATOGRAM: As the sample elutes from the column, a UV detector (typically set at 214nm or 220nm for peptide bond absorption) measures the components in real time, generating a chromatogram — a time-versus-absorbance graph. The primary, centralized peak represents the target peptide sequence. The integrated area of this peak, calculated as a percentage of the total area of all detected peaks, defines the purity percentage.
A clinically viable peptide should exhibit a sharp, symmetrical main peak representing greater than 98% of the total integrated area, with only minimal, well-resolved secondary peaks. Broad, asymmetric peaks or significant shoulder peaks indicate co-eluting impurities that the purification process failed to remove. Peak tailing suggests column overloading or secondary chemical interactions. Multiple peaks of comparable height suggest the sample contains a mixture of peptide species — a serious quality failure.
Mass Spectrometry (MS): Confirming Molecular Identity
While HPLC quantifies how much of the sample is the dominant species, Mass Spectrometry confirms what that species actually is. It is entirely possible to synthesize a 99% pure peptide that is the completely wrong amino acid sequence — a deletion sequence, an enantiomeric substitution, or even an entirely different molecule. MS prevents this catastrophic error.
MS utilizes precisely calibrated electromagnetic fields to measure the exact molecular weight (MW) of the compound, typically via Electrospray Ionization (ESI) or Matrix-Assisted Laser Desorption/Ionization Time-of-Flight (MALDI-TOF). The empirically measured molecular weight must align precisely with the theoretical molecular weight calculated from the peptide's molecular formula — typically within a strict tolerance of plus or minus 1 Dalton (Da).
For example, if evaluating BPC-157 (Theoretical MW: 1419.53 Da), a rigorous Certificate of Analysis will display an MS reading of approximately 1419.5 Da, verifying sequence identity. If the MS reading is off by more than 2 Da, you either have the wrong peptide, a substituted sequence, or a degraded product. Some advanced laboratories also provide MS/MS (tandem mass spectrometry) data, which fragments the peptide and confirms the actual amino acid sequence order — the highest level of identity confirmation available.
Bacterial Endotoxin Testing (LAL Assay): The Test Most Vendors Skip
This is the test that separates legitimate pharmaceutical-grade suppliers from gray market vendors — and it is the single most overlooked quality metric in the peptide community. The Limulus Amebocyte Lysate (LAL) assay detects bacterial endotoxins (lipopolysaccharides shed from the outer membrane of Gram-negative bacteria) at extraordinarily low concentrations — often below 0.25 Endotoxin Units per milliliter (EU/mL).
Why does this matter? Because endotoxins are not destroyed by standard sterilization techniques. You can autoclave, filter, or gamma-irradiate a peptide solution and kill every living bacterium — but the endotoxins those bacteria already shed into the solution remain biologically active and intact. When injected, even trace endotoxin levels trigger a violent innate immune cascade: the toll-like receptor 4 (TLR4) pathway activates, releasing TNF-alpha, IL-1-beta, IL-6, and other pro-inflammatory cytokines. At low levels, this manifests as unexplained fever, injection site swelling, and malaise. At higher levels, it can produce septic shock.
The FDA limit for parenteral drugs is 5 EU per kilogram of body weight per hour. For a 75 kg individual, that is 375 EU total — seemingly generous, but peptide solutions contaminated during non-sterile manufacturing can contain thousands of EU/mL. If a vendor does not provide LAL endotoxin test results on their Certificate of Analysis, they have not tested for it. Period. This is the single most important safety test for any injectable peptide, and the vast majority of gray market vendors do not perform it.
Purity vs. Peptide Content: The Critical Distinction
A widespread misconception in peptide analysis is equating Purity with Peptide Content. They are entirely separate analytical parameters measuring fundamentally different things:
PURITY (HPLC): The percentage of the peptide component that correctly matches the target sequence, relative to other peptide-like impurities. If the HPLC reads 99%, it means 99% of the peptide species in the vial are the correct sequence. The remaining 1% are truncated, oxidized, or otherwise modified sequences.
NET PEPTIDE CONTENT (NPC): The percentage of the total lyophilized (freeze-dried) mass in the vial that is actual peptide, as opposed to non-peptide components such as counter-ions (TFA or acetate salts from the purification process), residual moisture, and bound solvents. Standard NPC for research-grade peptides ranges from 60% to 85%, depending on the peptide sequence and its counter-ion load. Shorter peptides with high TFA content may have NPC as low as 50%.
THE CLINICAL IMPLICATION: If you possess a 5mg vial of peptide with a 99% HPLC purity but only a 75% Net Peptide Content, the vial contains exactly 3.75mg of the active therapeutic agent — not 5mg. Precision dosing calculations must account for net peptide content, not merely labeled vial weight. This distinction becomes clinically critical at microgram-level doses where the margin between therapeutic and sub-therapeutic concentrations is narrow.
The China Supply Chain: Understanding Where Your Peptides Come From
An estimated 90% or more of research-grade peptides sold globally originate from contract synthesis facilities in China — primarily concentrated in the Hubei, Anhui, Zhejiang, and Guangdong provinces. This is not inherently problematic: China possesses the world's largest installed base of solid-phase peptide synthesis (SPPS) equipment, highly trained peptide chemists, and the raw material supply chain (Fmoc-protected amino acids, resins, coupling reagents) necessary for large-scale peptide production.
The problem is not where the peptides are made — it is the variance in quality control standards across the hundreds of small and mid-size synthesis houses operating in this space. The market ranges from ISO-certified, NMPA-inspected GMP facilities producing clinical-trial-grade material, down to basement-level operations running single SPPS synthesizers with zero quality documentation.
WHAT DISTINGUISHES A QUALITY CHINESE MANUFACTURER: Facility-level certifications (ISO 9001, ISO 13485, NMPA registration); in-house analytical equipment (HPLC, LC-MS, LAL testing apparatus); batch-specific documentation with raw chromatogram data; sterile lyophilization suites with environmental monitoring; documented cold-chain shipping procedures; and willingness to provide samples for independent third-party verification. If a supplier cannot provide facility-level credentials or refuses independent testing, the price discount is not worth the risk.
THE REPACKAGING PROBLEM: Many western-facing peptide vendors do not synthesize their own products. They purchase bulk lyophilized powder from Chinese synthesis houses, repackage it into branded vials, and generate their own Certificates of Analysis — sometimes without ever testing the material they are selling. This is where the supply chain breaks down catastrophically. The vendor's CoA may show 99% purity, but if that CoA is based on a reference sample tested months ago from a different batch, it tells you nothing about the powder in your specific vial.
TikTok, Social Media, and the Peptide Misinformation Epidemic
The explosion of peptide content on TikTok, Instagram Reels, and YouTube Shorts has created a dangerous paradox: unprecedented public awareness of peptide therapy coupled with catastrophically low information quality about product sourcing and verification. Influencers routinely promote specific peptide vendors with affiliate discount codes while demonstrating zero understanding of analytical chemistry, sterility, or quality verification.
THE MOST COMMON SOCIAL MEDIA RED FLAGS: Vendors who only exist on social media platforms (no professional website, no physical address, no verifiable business registration). Influencers who claim to have "lab-tested" products but cannot produce batch-specific Certificates of Analysis with both HPLC and MS data. Vendors who show a single CoA repeatedly for different batches, different peptides, or over months of sales. Prices that are dramatically below market rate — legitimate peptide synthesis with proper purification (preparative HPLC), lyophilization, sterility testing, endotoxin testing, and cold-chain shipping has irreducible costs. If a vendor is selling 5mg of a complex peptide for eight dollars, something critical has been skipped.
THE AESTHETIC PACKAGING TRAP: A professional-looking label, a holographic anti-counterfeit sticker, and a QR code that links to a generic PDF do not constitute quality verification. The peptide industry has no mandatory packaging standards — anyone can purchase professional capping machines, custom labels, and pharmaceutical-grade glass vials from Alibaba for pennies per unit. Judge peptides by their analytical documentation, not their packaging.
WHAT INFORMED CONSUMERS DO INSTEAD: They request batch-specific CoAs before purchasing. They verify that the CoA includes both HPLC chromatogram data and Mass Spectrometry confirmation. They check for bacterial endotoxin (LAL) test results. They cross-reference CoA lot numbers with the lot number printed on the received vial. And increasingly, they submit samples to independent third-party laboratories for blind verification.
Independent Third-Party Testing: The Gold Standard of Verification
The most reliable way to verify peptide quality is to bypass the vendor entirely and submit a sample directly to an independent analytical laboratory. Several laboratories have emerged that cater specifically to the peptide research community, including Janoshik Analytical (Czech Republic), Vanguard Laboratory, and various ISO 17025-accredited contract testing facilities.
HOW THIRD-PARTY TESTING WORKS: You send a small sample (typically 1-2mg of lyophilized powder) to the laboratory with no identifying information about the vendor. The lab performs blind HPLC purity analysis, mass spectrometry identity confirmation, and (if requested) bacterial endotoxin screening and heavy metals testing. Results are typically returned within 5 to 14 business days.
WHAT TO TEST FOR: At minimum, request HPLC purity and MS identity confirmation. For injectable peptides, also request LAL endotoxin testing. For peptides from unknown or new suppliers, consider adding residual solvent screening (GC-MS) and heavy metals panels (ICP-MS for lead, arsenic, mercury, cadmium). The cost of comprehensive third-party testing typically ranges from $50 to $200 per sample — a small price relative to the cost of injecting a contaminated or misidentified compound.
GROUP TESTING AND COMMUNITY VERIFICATION: Online peptide research communities have pioneered a group-funded testing model where members pool resources to test specific batches from popular vendors. This creates crowdsourced quality data that benefits the entire community. While not a substitute for testing your own specific vial, community testing data provides valuable signal about a vendor's overall quality trajectory over time.
Evaluating a Certificate of Analysis (CoA): The Complete Red-Flag Checklist
A legitimate, rigorous Certificate of Analysis is the primary quality document in the peptide supply chain. However, the prevalence of fraudulent, generic, and misleading CoAs has made verification a critical skill. Here is what to verify — and what raises immediate red flags:
REQUIRED ELEMENTS OF A LEGITIMATE CoA: A unique lot or batch number that matches the number on your specific vial or container. The peptide name, molecular formula, molecular weight, and CAS number (if applicable). HPLC purity results expressed as a percentage of main peak area, with the actual chromatogram image included (not just a number). Mass Spectrometry results showing the observed molecular weight versus the theoretical molecular weight (within plus or minus 1 Da tolerance). Testing date that aligns with the manufacturing date of your batch. Name and credentials of the quality control analyst or laboratory director who signed the document. The name, address, and contact information of the testing laboratory.
RED FLAGS THAT INDICATE A FRAUDULENT OR UNRELIABLE CoA: A CoA that lists purity as exactly 99.0% or 99.9% with no chromatogram data — legitimate HPLC results always come with the raw chromatogram image. A document that looks like a generic template with the peptide name and purity number simply typed in. No lot or batch number, or a lot number that does not match the vial you received. A CoA that is months or years old — peptide quality degrades over time, and a CoA from a different production batch is meaningless. A testing laboratory that cannot be independently verified (no website, no business registration, no published accreditations). A CoA that shows only HPLC data without Mass Spectrometry — you cannot confirm identity from purity alone. Missing endotoxin testing for injectable peptides.
Purity Tiers: What the Numbers Actually Mean
Not all purity levels are created equal, and different applications demand different minimum thresholds:
GREATER THAN 99% (Pharmaceutical / Clinical-Trial Grade): This is the standard for FDA-approved peptide drugs (semaglutide, tirzepatide) and clinical trial investigational materials. Achieving this requires multiple rounds of preparative HPLC purification, stringent environmental controls, and comprehensive analytical validation. Cost-prohibitive for most research applications.
98% TO 99% (Research-Grade Premium): The gold standard for high-quality research peptides. Suitable for rigorous pharmacokinetic studies and the standard expected by serious researchers. Indicates effective synthesis and thorough purification. This is the minimum threshold recommended by CalcMyPeptide for any injectable research application.
95% TO 98% (Research-Grade Standard): Acceptable for many research applications, particularly in vitro cell culture studies and preliminary dose-finding experiments. The 2-5% impurity fraction is unlikely to cause acute safety issues at standard research doses but may introduce variability in results.
90% TO 95% (Economy / Screening Grade): Suitable only for initial screening, binding assays, and non-injectable applications. The impurity burden at this level may include biologically active truncated sequences that confound experimental results. Not recommended for any in vivo application.
BELOW 90% (Crude / Unacceptable): Indicates failed synthesis, inadequate purification, or advanced degradation. Material at this purity level should not be used for any research application. A vendor selling sub-90% peptides as research-grade is either incompetent or fraudulent.
Storage, Handling, and Post-Purchase Degradation
Even a perfectly synthesized, laboratory-verified peptide will degrade if handled improperly after purchase. The primary degradation pathways are hydrolysis (water-mediated bond cleavage), oxidation (particularly of methionine and tryptophan residues), deamidation (asparagine and glutamine converting to aspartate and glutamate), and microbial contamination of reconstituted solutions.
LYOPHILIZED (UNRECONSTITUTED) PEPTIDES: Store at minus 20 degrees Celsius or below for long-term stability (years). Protect from light, moisture, and repeated freeze-thaw cycles. Desiccant packets in the storage container help absorb ambient moisture. At room temperature, most lyophilized peptides remain stable for weeks to months — but this is the riskiest storage condition.
RECONSTITUTED PEPTIDES: Once reconstituted with bacteriostatic water (BAC water containing 0.9% benzyl alcohol as a preservative), store at 2 to 8 degrees Celsius (standard refrigerator temperature). Use within 28 days. Do NOT freeze reconstituted peptides — the freeze-thaw cycle can denature the peptide structure and cause aggregation, which reduces bioactivity and may increase immunogenicity.
CRITICAL HANDLING RULES: Always use bacteriostatic water for multi-dose reconstitutions — sterile water lacks preservative and becomes a bacterial growth medium after the first needle puncture. Never shake a reconstituted peptide vial — this causes foaming and denaturation at the air-liquid interface. Roll or swirl gently. Use insulin syringes with fresh needles for every draw to maintain sterility. Label every vial with the reconstitution date, peptide name, and concentration.