The FDA-Approved Visceral Fat Protocol
Unlike the vast majority of peptides utilized in the optimization space, Tesamorelin operates under a distinct regulatory and clinical paradigm: it is fully FDA-approved (under the brand name Egrifta SV) specifically for the reduction of excess visceral adipose tissue (VAT).
Tesamorelin is a synthetic 44-amino acid analogue of Growth Hormone-Releasing Hormone (GHRH). By binding to receptors on the anterior pituitary, it forces a massive, pulsatile release of endogenous growth hormone. What separates Tesamorelin from other GHRH analogues (like CJC-1295) is its aggressive, targeted affinity for lipolyzing visceral fat—the hard, inflammatory, metabolically destructive fat that wraps around internal organs.
The 26-Week Time Horizon
The single most common error in Tesamorelin administration is premature cycle cessation. The pharmacokinetics of GHRH analogues dictate a significant delay between biochemical receptor activation (which happens instantly) and macroscopic morphological changes (fat loss).
According to the pivotal Phase III clinical trials governing its FDA approval, elevated IGF-1 is detectable within the first 7 days, peaking around Week 4. However, clinically significant changes in Visceral Adipose Tissue (VAT) do not register on CT scans until Week 12. Maximum therapeutic fat reduction (-15.2% visceral trunk fat on average) requires a continuous 26-week minimum administration protocol.
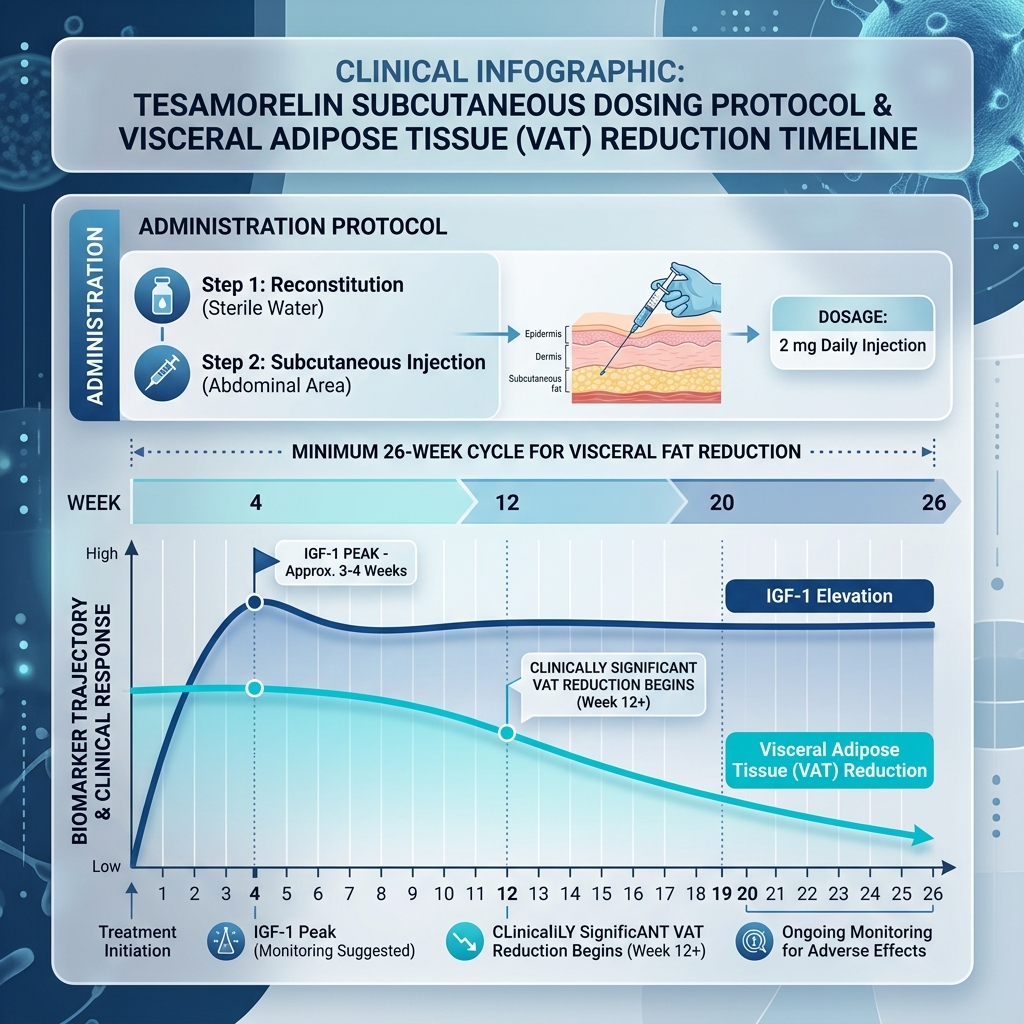
Clinical Dosing Parameters
• The Clinical Dose: The FDA-approved protocol utilizes a static dose of 2mg (2000mcg) per day. Because of its short half-life (approx. 26 minutes), this creates a powerful, physiological spike mimicking a natural GH pulse.
• Administration: Subcutaneous injection into the abdomen, rotating sites daily to avoid lipohypertrophy.
• Timing Protocol: Fasting is mandatory. Inject once daily, ideally in the morning upon waking (waiting 60 minutes before caloric intake) or directly before bed (fasting for a minimum of 2-3 hours prior). Insulin completely blunts GHRH-mediated growth hormone release.
• Mandatory Monitoring: Baseline IGF-1 and HbA1c panels must be drawn prior to initiation. At Week 6, re-test IGF-1. If levels exceed the upper limit of the age-adjusted reference range, the dose must be titrated downward to avoid acromegalic or insulin-resistant side effects.