What is Bacteriostatic Water?
Bacteriostatic water (BAC water) is sterile water for injection containing 0.9% benzyl alcohol as a bacteriostatic preservative. The benzyl alcohol inhibits bacterial growth by disrupting microbial cell membrane integrity, allowing the vial to be punctured multiple times over a 28-day window without contamination.
It is the standard reconstitution solvent for all multi-dose peptide vials. Using plain sterile water instead eliminates this protection, requiring single-use within 24 hours. The clinical consensus strictly dictates the use of bacteriostatic water for any compounded sub-q agent intended for repeated dosing.
Beyond simple sterility, the 0.9% benzyl alcohol content serves a highly critical role. It operates as an antimicrobial agent that halts the division of bacteria rather than killing them outright (hence "bacteriostatic" rather than "bactericidal"). Over the typical 28-day clinical timeframe, this is entirely sufficient to maintain the purity of the peptide solution within the vial, provided the user exercises sound aseptic techniques during extraction.
Aseptic Technique and Needle Contamination
The preservation power of bacteriostatic water is not absolute. Proper aseptic technique is mandatory. This begins with swabbing both the bacteriostatic water vial stopper and the peptide vial stopper with 70% isopropyl alcohol before every puncture.
Furthermore, drawing from the bacteriostatic water vial with a dull or previously used needle can introduce foreign matter and drastically compromise the integrity of the solution. Every extraction must utilize a completely sterile, brand new syringe and needle. The stopper itself, typically constructed from pharmaceutical-grade halobutyl rubber, is designed to self-seal after puncturing. However, using needles that are too thick (like 18G drawing needles) can cause "coring"—where a tiny piece of the rubber is carved out and falls into the solution, rendering it compromised.
It is generally recommended to use 27G to 31G insulin syringes for extraction to minimize trauma to the stopper.
Expiration and Storage Dynamics
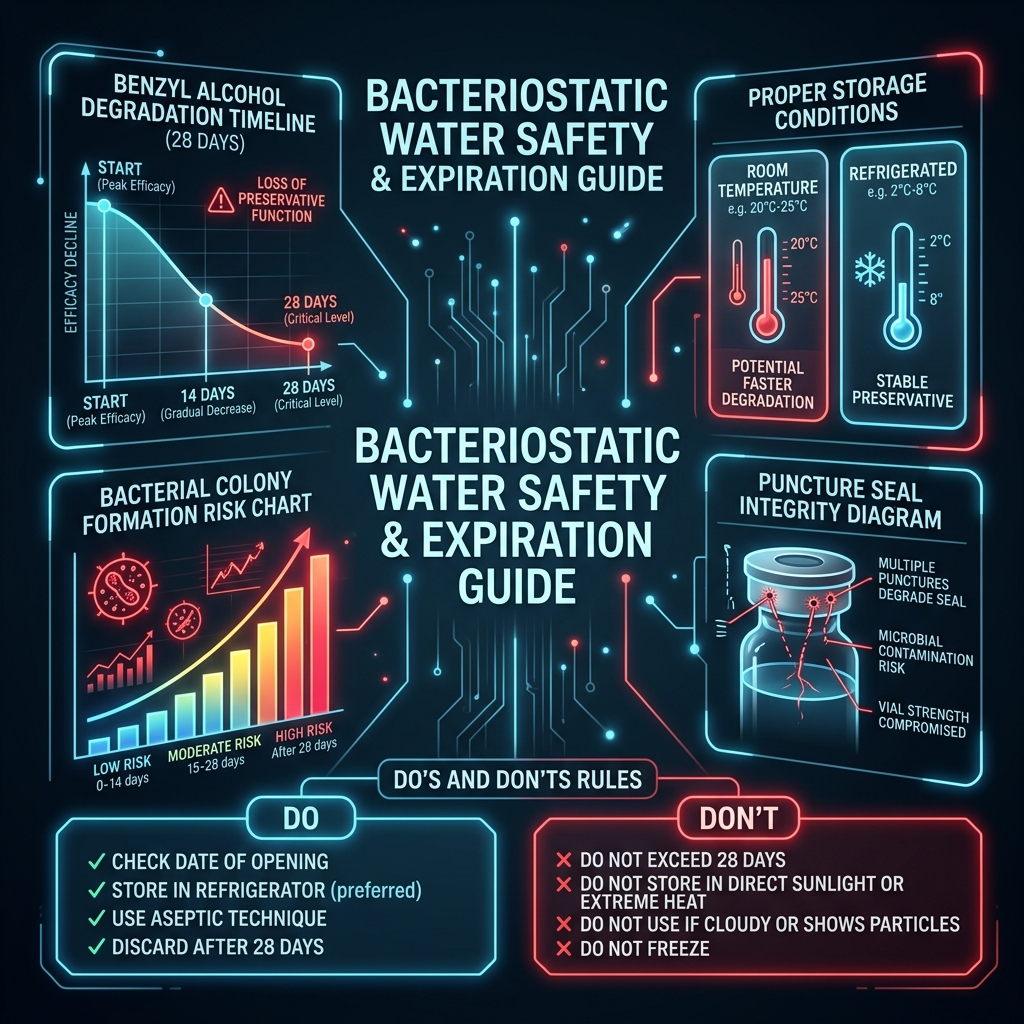
Unopened BAC water has a manufacturer's shelf life of typically 2 to 3 years when stored at room temperature (20-25°C) away from direct UV light. The exact expiration date is strictly printed on the vial surface.
Once punctured, the FDA-mandated 28-day expiration clock immediately begins. Why exactly 28 days? Over time, and with repeated exposure to the air within the headspace of the vial and the rubber stopper interaction, the 0.9% benzyl alcohol concentration degrades. As the concentration of benzyl alcohol drops, the fluid loses its ability to reliably inhibit bacterial growth.
After 28 days, the risk curve increases exponentially. Discard any opened BAC water vial after 28 days regardless of how much liquid remains in the vial. A surprisingly common error is patients trying to use a single 30mL vial of bacteriostatic water for 6 months because they are only reconstituting small 5mg peptide vials. This practice introduces massive systemic infection risk. Always chart the puncture date on the vial label with a marker.
Upon constitution of the peptide, the mixed solution should typically be stored upright in the refrigerator to protect the peptide bonds from heat degradation, although the bacteriostatic water itself (before mixing) is completely comfortable at room temperature.
Are All Waters The Same? (Sterile Water vs. BAC vs. Sodium Chloride)
It is a frequent source of confusion: differentiating between Bacteriostatic Water, Sterile Water for Injection, and Bacteriostatic Sodium Chloride. Sterile Water contains zero preservatives. It is generally used for single-dose intravenous mixtures that are administered immediately. Using plain sterile water for a multi-use peptide vial is a direct route to bacterial blooming.
Bacteriostatic Sodium Chloride (0.9% NaCl with Benzyl Alcohol) is sometimes used for specific drugs, but it introduces an osmotic solute (salt) into the suspension. Certain sensitive peptides can actually denature or crash out of solution upon interacting with the chloride ions. Therefore, standard Bacteriostatic Water for Injection (H2O + 0.9% Benzyl Alcohol) is the universal, absolute gold standard for reconstituting heavily structured peptides like Semaglutide, Tirzepatide, BPC-157, and GH secretagogues.