Categorical Adverse Event Framework
In clinical peptide therapy, there is no such thing as a "side effect"—there are only primary biological mechanisms and subsequent systemic adaptations. No peptide is completely inert.
Unlike single-target pharmaceuticals, peptides often modulate upstream regulatory pathways, meaning their secondary effects vary dramatically between peptide classes. A GLP-1 receptor agonist presents an entirely different adverse event profile than a GH secretagogue or an angiogenic healing peptide.
This guide categorizes adverse events (AEs) by distinct peptide class, categorizing them from highly prevalent adaptations to rare, severe contraindications that demand immediate clinical protocol cessation.
GLP-1 Receptor Agonists (Semaglutide, Tirzepatide, Retatrutide)
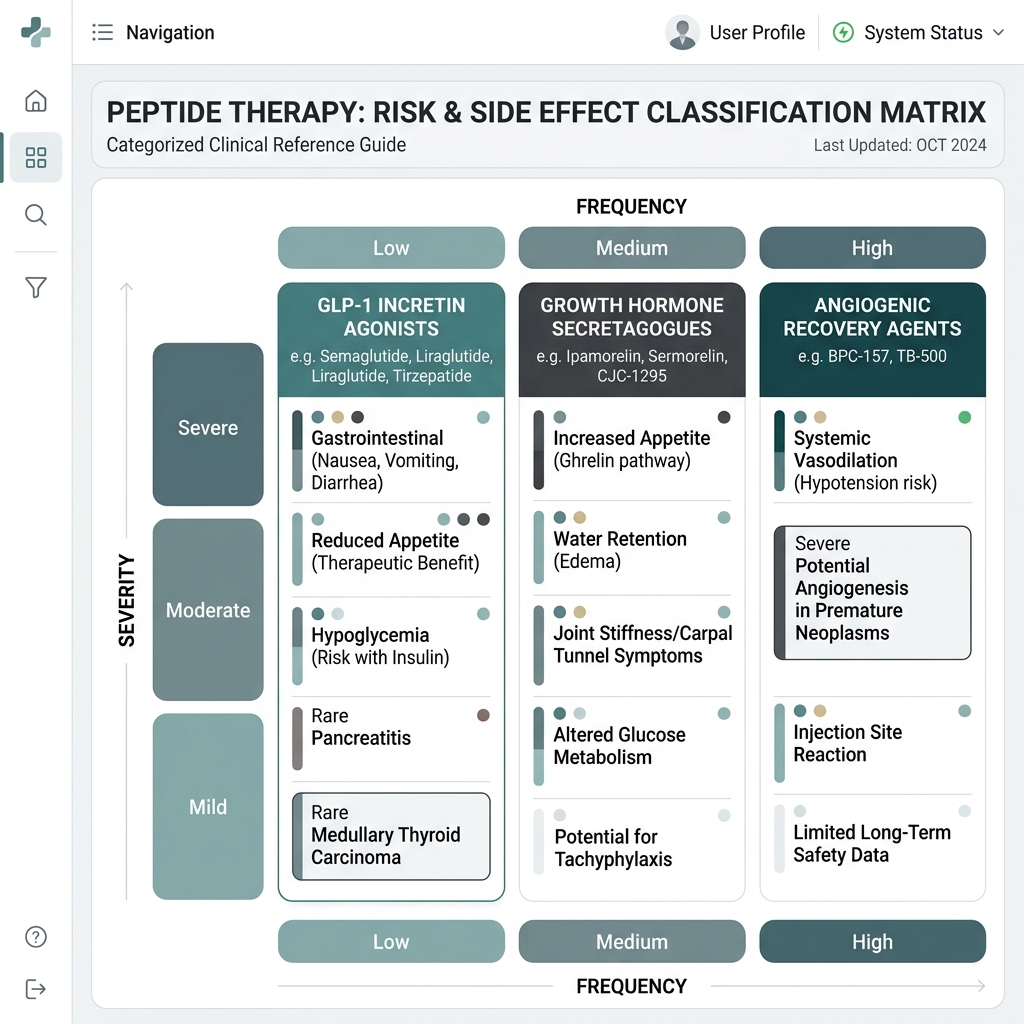
GLP-1 agonists fundamentally alter gastric emptying and central appetite signaling. Consequently, their adverse event profile is heavily gastrointestinal.
• High Prevalence (>10%): Nausea (particularly acute during up-titration phases), transient diarrhea, constipation, profound appetite suppression leading to caloric deficit fatigue, and mild vomiting. These physiological adaptations generally resolve within 4–6 weeks at a stable dose.
• Moderate Prevalence (1-10%): Localized injection site reactions, headaches secondary to dehydration, orthostatic dizziness, generalized fatigue, and exacerbation of existing gastroesophageal reflux disease (GERD).
• Rare but Critical (<1%): Acute pancreatitis, gallbladder disease (cholelithiasis/cholecystitis), and clinically significant hypoglycemia (particularly when co-administered with exogenous insulin or sulfonylureas).
Absolute Contraindications: Personal or familial history of Medullary Thyroid Carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Active or historical pancreatitis.
Growth Hormone Secretagogues (Ipamorelin, CJC-1295, MK-677)
These compounds drive pituitary somatotrophs to release endogenous Growth Hormone. Their side effects are directly correlated to downstream increases in IGF-1 and systemic fluid retention.
• High Prevalence: Extracellular water retention (presenting as puffiness in the extremities/face), significantly increased hunger (specifically with ghrelin mimetics like GHRP-6 and MK-677), and vivid, hyper-lucid REM sleep states.
• Moderate Prevalence: Transient numbness or tingling in extremities (frequently mimicking carpal tunnel syndrome due to fluid compression on nerves), joint stiffness, daytime somnolence, and an acute head rush immediately post-injection (common with CJC-1295/Mod GRF 1-29).
• Rare but Critical: Exacerbation of insulin resistance (monitor fasting glucose), and clinically significant elevations in cortisol and prolactin (seen with older GHRPs like Hexarelin and GHRP-2, but notably absent with Ipamorelin).
Absolute Contraindications: Active oncological processes (as elevated GH/IGF-1 can accelerate tumor proliferation), severe unmanaged diabetes, and compressive neuropathies.
Angiogenic Healing Peptides (BPC-157, TB-500)
Repair peptides function by rapidly upregulating cellular migration and new blood vessel formation. They are generally considered to have the highest tolerability profile in the peptide landscape.
• High Prevalence: Mild, transient localized erythema (redness) or edema (swelling) at the subcutaneous injection site.
• Moderate Prevalence: Transient orthostatic hypotension immediately following administration, acute mild nausea, and generalized lethargy.
• Theoretical Oncological Risk: Because BPC-157 and TB-500 aggressively promote angiogenesis (via VEGF and FGF pathways), they could theoretically accelerate vascular supply to existing, undiagnosed malignant tumors. While not definitively proven in human models, this remains a significant clinical caution.
Absolute Contraindications: Known active malignancies or a high-risk history of cancer within the preceding five years. Pregnancy or breastfeeding.
Cognitive & Mitochondrial Regulators (Dihexa, SS-31, MOTS-c)
Nootropic and mitochondrial peptides operate on specialized neurological and metabolic pathways, presenting highly unique adverse profiles.
• Nootropics (Selank/Semax): Often administered intranasally; can cause rhinitis/nasal mucosal irritation, mild sedation (Selank), subtle shifts in dopaminergic sensitivity, and transient blood pressure fluctuations.
• Mitochondrial (MOTS-c/SS-31): May induce acute shifts in fasting blood glucose (MOTS-c mimics high-intensity exercise metabolism) and localized injection site inflammation.
• The Dihexa Caution: Dihexa exhibits extreme potency in driving synaptogenesis. Dosage ceilings and long-term neuro-architectural side effects remain poorly established in human clinical models. Proceed with exceptional caution.
Clinical Red Flags: Immediate Protocol Cessation
Immediate medical evaluation is required, and peptide administration must be halted, if an individual experiences:
• Acute, severe abdominal pain radiating to the back (indicative of GLP-1 induced pancreatitis).
• Systemic anaphylaxis: urticaria (hives), facial/airway edema, or acute dyspnea.
• Intractable nausea resulting in an inability to maintain hydration.
• Neurological or visual field changes (particularly relevant during GH secretagogue protocols).
• Deep, expanding erythema accompanied by purulence or extreme heat at an injection site (severe infection).
Taper or Discontinue For: Unresolving systemic edema, progressive fasting hyperglycemia, or neuropathic pain (numbness/tingling) that escalates despite dose titration.