Understanding Lyophilization
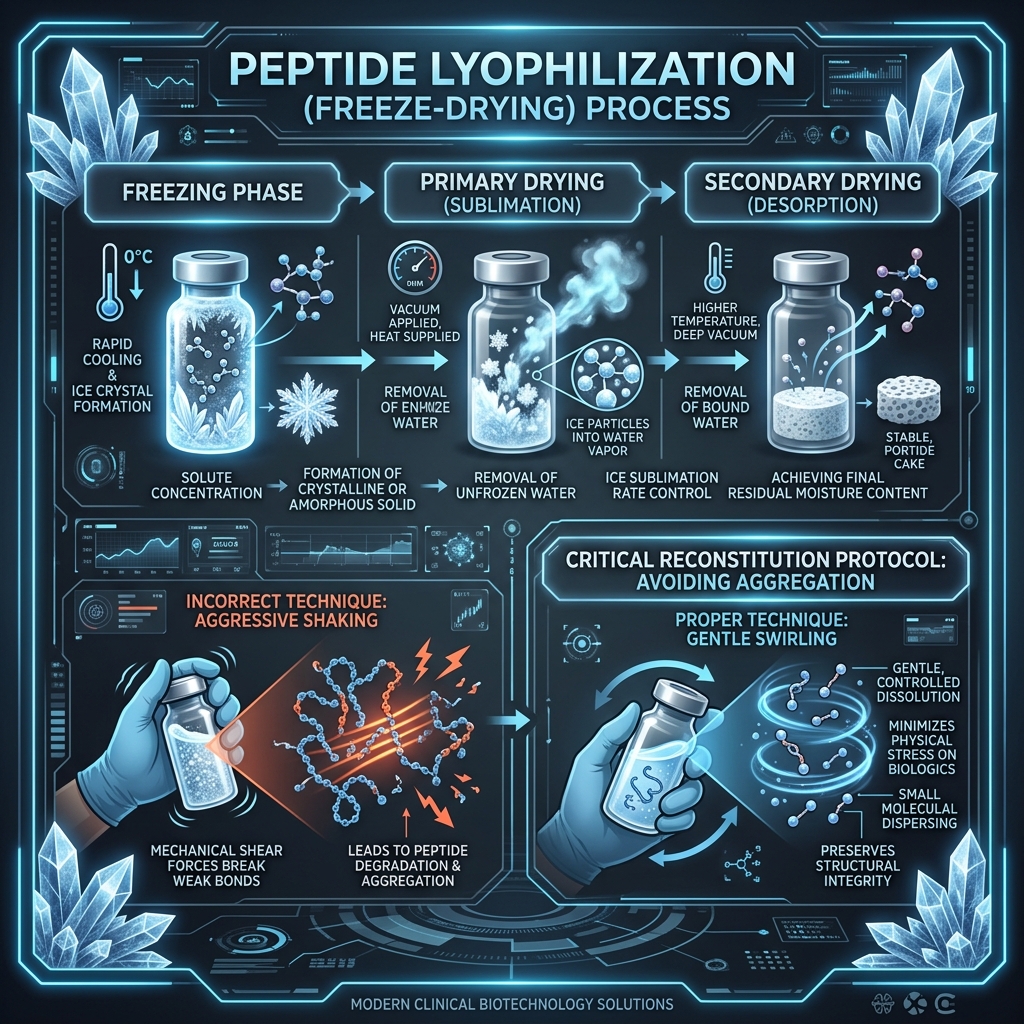
Lyophilization, commonly known as freeze-drying, is the highly specialized process that converts liquid peptide solutions into a stable, shelf-storable powder known as a "puck" or "cake." In the pharmaceutical and compounding industries, this is universally utilized because peptides suspended in an aqueous solution are inherently unstable and subject to rapid enzymatic and hydrolytic degradation.
The process begins by deep-freezing the peptide solution to extreme temperatures, often -40°C or colder. Once completely frozen, the vials are placed into a specialized vacuum chamber. Under intense negative pressure, the ambient temperature is very slightly raised, causing the frozen water (ice) to undergo sublimation—shifting directly from a solid state to water vapor, completely bypassing the liquid phase. This vapor is then drawn out and collected by condenser coils.
What remains behind is a highly porous, sponge-like matrix of pure, sterile peptide molecules. Because the water has been removed without the use of boiling heat, the delicate three-dimensional folding structure of the peptide chains remains intact. Properly lyophilized peptides, sealed in a vacuum or under inert nitrogen gas, can maintain greater than 95% potency for months at room temperature, and easily exceed two years of total stability when stored in a standard -20°C freezer.
The Danger of Heat Degradation
While lyophilization provides exceptional stability compared to a mixed liquid solution, it is not invincible. Ambient environmental heat is the primary enemy of dry peptides. Temperatures exceeding 40°C (104°F) can trigger localized thermal agitation sufficient to break weaker non-covalent interactions within the peptide's tertiary structure. Over sustained periods, thermal energy initiates peptide bond hydrolysis even in the absence of liquid water.
During the height of summer, a package sitting inside a metal delivery truck can easily reach 130°F. If lyophilized peptides are subjected to this heat continuously for several days, substantial degradation can occur, leading to decreased drug efficacy or inactive fragments. Top-tier pharmacies counter this by utilizing insulated packaging and frozen thermal packs during transit. For ultimate longevity, end-users should transfer vials to frozen storage (-20°C) immediately upon arrival.
Mechanical Agitation: The "Never Shake" Rule
In both their liquid and lyophilized states, peptide chains are surprisingly fragile when it comes to mechanical shear forces. Once you have introduced bacteriostatic water back into the vial (reconstitution), shaking or vortexing the mixture creates thousands of microscopic air-water interfaces (bubbles).
The surface tension forces present at these microscopic air-water boundaries are intense enough to physically rip long amino acid chains apart, unfolding their structures and permanently destroying their biological activity. This phenomenon is known as surface-induced denaturation. This is the precise reason why reconstitution instructions universally mandate: "Swirl gently, never shake."
Interestingly, this fragility extends partially to the dry, freeze-dried state as well. Severe mechanical agitation during shipping—such as dropping the box multiple times or intense vibration—can cause the beautifully structured lyophilized "cake" to physically shatter into dust. While a shattered cake does not automatically mean the peptide is ruined (as long as it wasn't subjected to heat), it does expose far more surface area to any residual moisture or oxygen that might be present in the vial's headspace. Thus, handling both wet and dry vials with reasonable care preserves maximum potency.
Proper Reconstitution Protocol
To correctly mix your peptide, aim the needle tip gently towards the glass inner wall of the vial, rather than spraying the bacteriostatic water forcefully straight down onto the fragile powder cake. Allow the water to cascade down the glass and smoothly contact the powder.
Once the water has been added, slowly roll the vial completely between your palms, or gently swirl it in a circular motion on a tabletop until the solution is completely clear and free of "floaters." Some highly dense peptides, like certain GHK-Cu formulations or larger chained analogues, may take 5-10 minutes of gentle swirling to fully dissolve. Be patient, avoid shaking, and store perfectly upright in the refrigerator post-reconstitution.