⚕️ Medical Disclaimer
⚕️ Medical Disclaimer: This article is for educational and informational purposes only. It does not constitute medical advice. Consult a qualified healthcare provider before using any peptide.
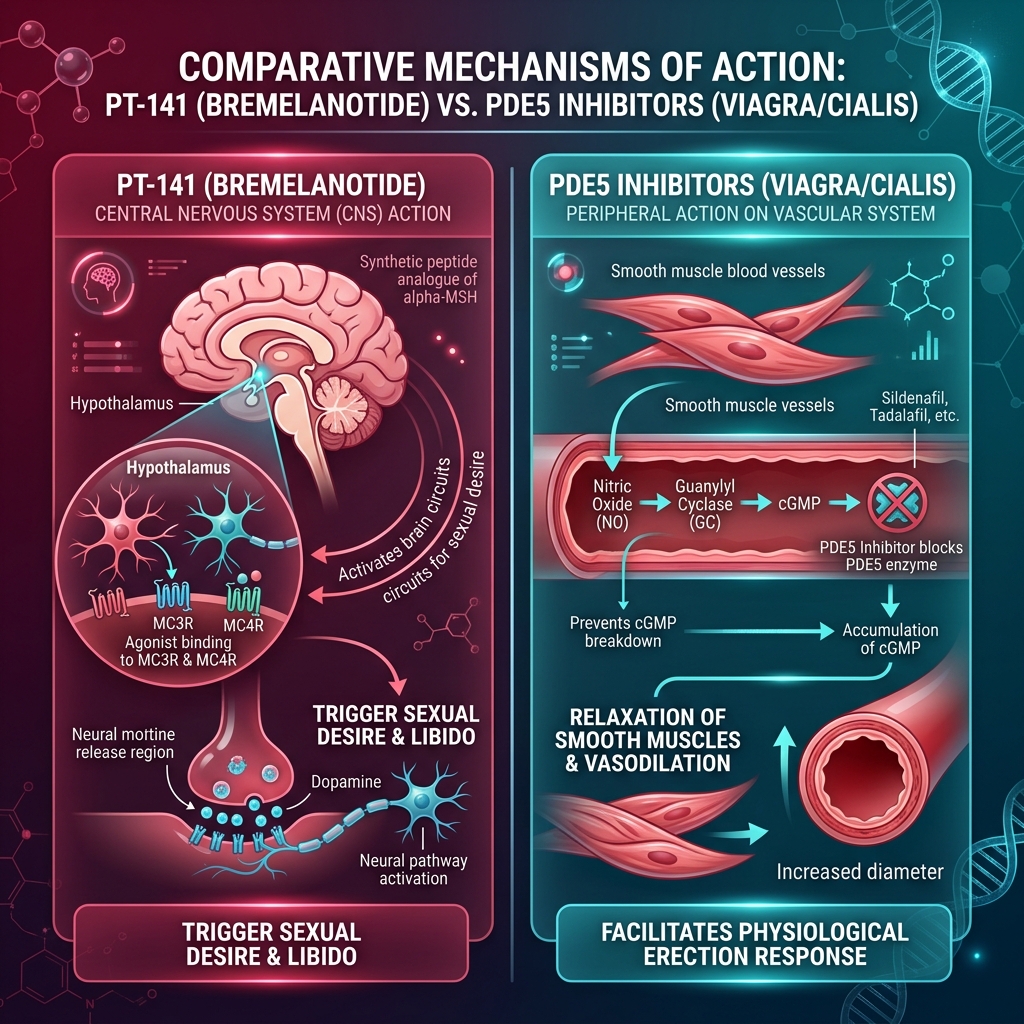
The Central Nervous System Paradigm of PT-141
For decades, the pharmacological treatment of sexual dysfunction has focused almost exclusively on the peripheral vascular system (i.e., PDE5 inhibitors like Viagra and Cialis). PT-141 (Bremelanotide) represents a paradigm shift. It is a synthetic melanocortin receptor agonist that bypasses the vascular system entirely, instead crossing the blood-brain barrier to act directly on the central nervous system.
Approved by the FDA under the brand name Vyleesi for Hypoactive Sexual Desire Disorder (HSDD) in premenopausal women, PT-141 is the only physiological aphrodisiac available in modern medicine. Instead of merely facilitating the mechanics of blood flow, it binds to the MC3r and MC4r receptors in the hypothalamus, actively triggering the neural pathways responsible for sexual drive, arousal, and libido in both men and women.
Clinical Dosing Parameters
• The Clinical Dose: The FDA-approved dose for women is 1.75mg (1750mcg), injected subcutaneously. Off-label use in men typically ranges from 1mg to 2mg. Doses exceeding 2mg do not linearly increase efficacy but exponentially increase the risk of severe nausea.
• Administration Timing: Unlike PDE5 inhibitors which take effect in 30-60 minutes, PT-141 has a profoundly delayed onset. It must be administered 2 to 4 hours prior to sexual activity. The effects are exceptionally long-lasting, frequently maintaining efficacy for 12 to 24 hours, and peaking at the 4-hour mark.
• Administration Limits: Clinical guidelines strictly warn against exceeding one dose within a 24-hour period, and recommend no more than 8 doses per month. Chronic, daily administration of melanocortin agonists can lead to receptor downregulation and permanent hyperpigmentation.
Side Effect Profile: Managing the Nausea Component
The primary limiting factor of PT-141 is not cardiovascular, but gastrointestinal. In clinical trials, up to 40% of patients experienced nausea following administration, typically peaking at the 1-2 hour mark and resolving by hour 4.
In high-end clinical practice, this nausea is frequently mitigated by co-administering an antiemetic such as Ondansetron (Zofran) 4-8mg approximately 30 minutes prior to the PT-141 injection. Additionally, transient increases in blood pressure (up to 6 mmHg systolic) were noted in trials; therefore, PT-141 is contraindicated in patients with uncontrolled hypertension.