Strategic Overview: The Era of Poly-Agonist Supremacy
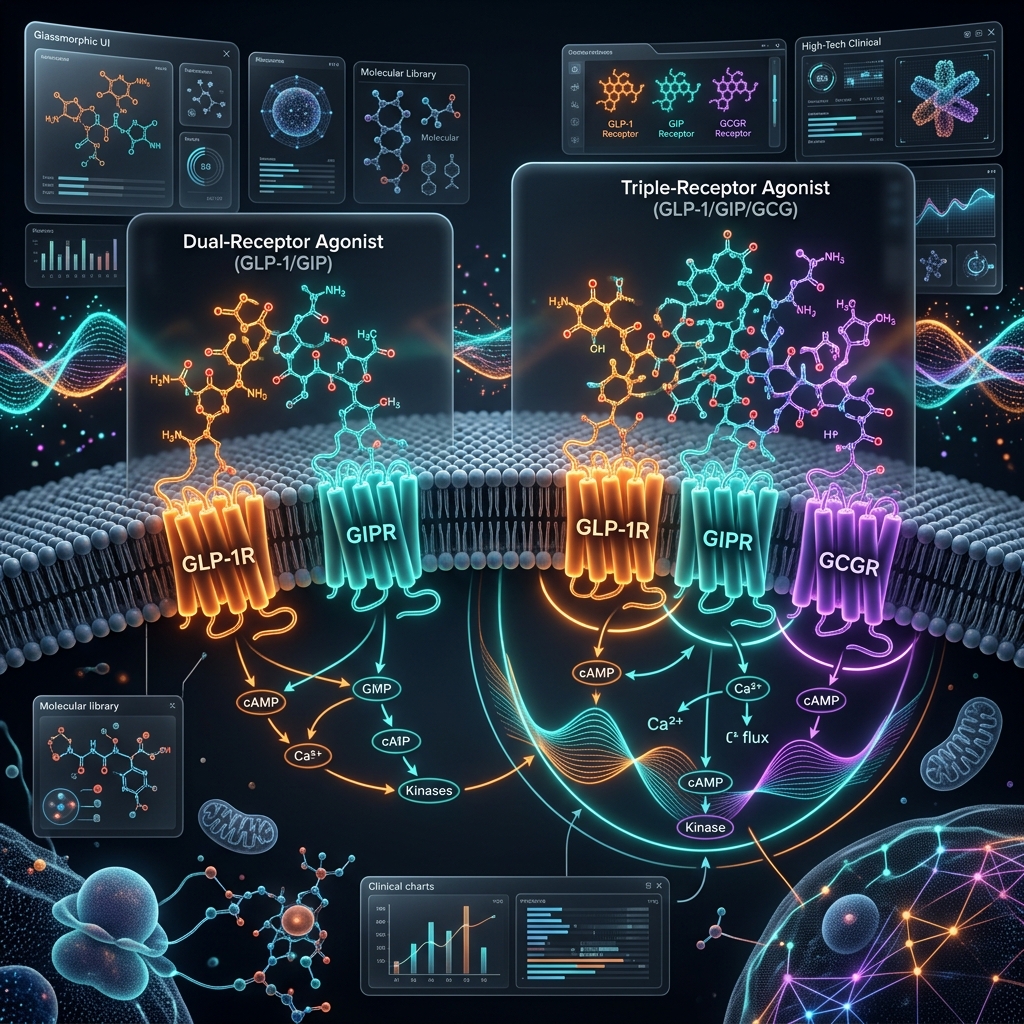
The introduction of Tirzepatide permanently altered the landscape of metabolic medicine. By engineering a dual-agonist capable of simultaneously binding to both the Glucagon-Like Peptide-1 (GLP-1) and Glucose-Dependent Insulinotropic Polypeptide (GIP) receptors, endocrinologists achieved a level of profound glycemic control and drastic adipose reduction historically reserved exclusively for highly invasive bariatric surgeries. However, the pharmacological frontier has already advanced. Retatrutide has emerged from advanced-stage clinical trials as the world’s first "Triple G" agonist—a synthetic leviathan designed to simultaneously target GLP-1, GIP, and crucially, the Glucagon receptor. The integration of active glucagon agonism fundamentally shifts the therapeutic paradigm from merely starving the system via massive appetite suppression to actively incinerating stored lipids by forcefully upregulating the basal metabolic rate.
Mechanism of Action: The Glucagon Variable
Tirzepatide operates essentially as a supreme defensive weapon: GLP-1 activation paralyzes gastric motility to induce intense, sustained satiety, while the GIP component acts centrally on the hypothalamus to further nullify cravings and dramatically amplify pancreatic insulin sensitivity. While this enforces a brutal, unbreakable caloric deficit, it inevitably leads to metabolic adaptation, where the body slows its resting energy expenditure to compensate for the perceived starvation.
Retatrutide bypasses this fatal adaptation. It retains the aggressive appetite suppression and insulin optimization of GLP-1/GIP, but introduces the Glucagon receptor as an offensive battering ram. Glucagon activation physically forces the liver to rapidly initiate glycogenolysis and hepatic lipolysis. Even while the patient is digesting standard meals, the peptide convinces the cellular mitochondria that the body is in a state of deep fasting. This continuous systemic demand for fuel drastically accelerates resting energy expenditure, turning the patient into a literal metabolic furnace.
Evaluating Phase 2 Clinical Trial Escalations
The aggregate data emerging from Phase 2 clinical trials has shattered all prior pharmacological benchmarks. At the culmination of a 48-week trial, the 12mg target dosage of Retatrutide produced an astonishing 24.2% mean reduction in total body weight. To contextualize this devastation of visceral fat: Tirzepatide (at its maximum 15mg clinical titration) achieved a 22.5% body weight reduction, but required an agonizing 72 weeks of administration to reach that absolute ceiling. Retatrutide is not simply marginally more effective; the velocity of its adipose destruction operates on a totally detached timeline.
Hepatic Reversal: Eradicating Non-Alcoholic Steatohepatitis
Perhaps the most profound clinical revelation associated with Retatrutide's glucagon synergy is its unprecedented impact on Non-Alcoholic Fatty Liver Disease (NAFLD) and severe steatohepatitis. The liver inherently possesses a massive density of glucagon receptors. Through aggressive hepatic lipolysis, Phase 2 trial participants on Retatrutide demonstrated near-complete systemic clearance of toxic liver fat—recording up to an 86% relative mass reduction. This positions Retatrutide not just as an elite cosmetic weight-loss agent, but as a potentially curative therapeutic intervention for patients staring down the barrel of late-stage metabolic liver failure—a clinical milestone that dual-agonists have simply failed to achieve at scale.
The Tolerability Tax: Managing Sympathetic Overdrive
This magnitude of extreme metabolic efficacy exacts a brutal toll on systemic tolerability. Retatrutide does not gently coax weight loss; it aggressively stimulates the sympathetic nervous system due to intense, unremitting glucagon activation. Trial patients document significantly elevated resting heart rates, transient cardiac arrhythmias, hyperesthesia (extreme touch-sensitivity of the skin over nerve clusters), and a dramatically steeper curve of debilitating nausea when compared to Tirzepatide. For clinical implementation, the titration schedules for Retatrutide must be exponentially more cautious, requiring constant cardiovascular monitoring to ensure the patient does not succumb to sympathetic exhaustion.