What Is Melanotan II?
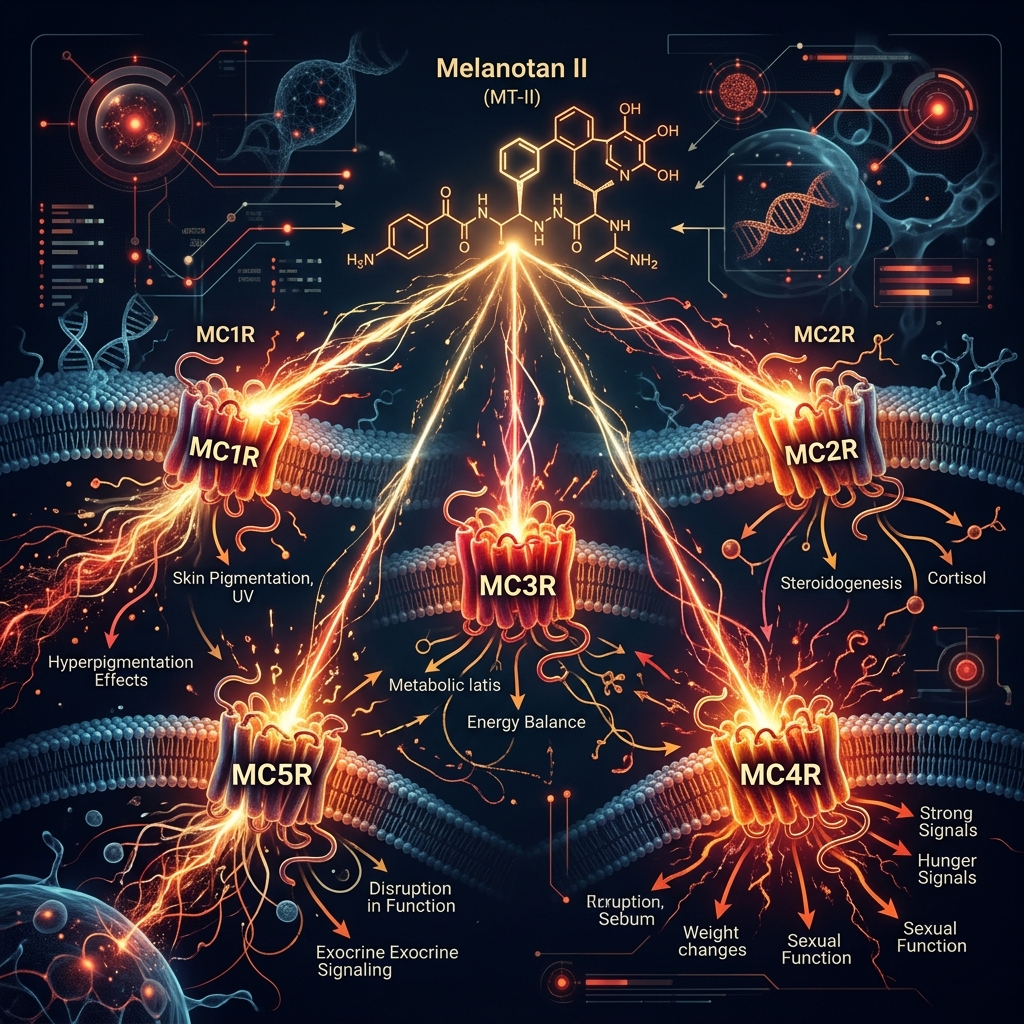
Melanotan II (MT-II) is a synthetic analog of alpha-melanocyte stimulating hormone (alpha-MSH) that stimulates melanogenesis — the production of melanin in the skin. It produces tanning without UV exposure, earning it the nickname "The Barbie Drug" on TikTok and Instagram.
MT-II binds to melanocortin receptors (MC1R through MC5R), which explains both its tanning effect and its array of side effects: MC1R drives melanogenesis, MC3R/MC4R affect appetite and sexual function, and MC5R affects sebaceous glands.
Why Dermatologists Are Alarmed
Nevi changes: MT-II stimulates melanocytes indiscriminately, including those in pre-existing moles. Multiple case reports document darkening, growth, and dysplastic changes in nevi following MT-II use. This complicates melanoma surveillance significantly.
Melanoma risk: While MT-II has not been proven to cause melanoma, it stimulates the same cellular pathways (melanocyte proliferation) that drive melanoma development. Using it in individuals with atypical mole syndrome or family history of melanoma is particularly concerning.
MT-II is not approved by any regulatory agency for tanning. All supplies come from the unregulated gray market with no quality guarantees.
Other Significant Risks
Cardiovascular: MT-II increases blood pressure through MC4R activation. Hypertensive episodes have been documented in case reports.
Sexual side effects: MC3R/MC4R activation causes spontaneous erections (priapism risk in males) and unpredictable libido changes. This is pharmacologically identical to how PT-141/bremelanotide works — which is FDA-approved specifically for this purpose.
GI effects: Nausea, flushing, and facial flushing are common (>30% of users), especially during the loading phase.
Fibrotic complications: Rare case reports of retroperitoneal fibrosis and rhabdomyolysis, though causality is not established.
The Bottom Line
Melanotan II provides a real tanning effect — the pharmacology works. But the risk profile is significant: nevi changes that complicate cancer screening, cardiovascular effects, and zero regulatory oversight on product quality.
Safer alternatives for tanning: DHA-based self-tanners (topical, no systemic effects), or controlled low-dose UV exposure with proper sunscreen on non-exposed areas.
Medical Disclaimer: This article is for educational purposes only. This is not an endorsement of Melanotan II. Consult a dermatologist before using any melanogenic agent.